- Review
- Open access
- Published:
Piperidine nucleus in the field of drug discovery
Future Journal of Pharmaceutical Sciences volume 7, Article number: 188 (2021)
Abstract
Background
Piperidine is an essential heterocyclic system and a pivotal cornerstone in the production of drugs. Piperidine byproducts showed several important pharmacophoric features and are being utilized in different therapeutic applications.
Main text
Piperidine derivatives are being utilized in different ways as anticancer, antiviral, antimalarial, antimicrobial, antifungal, antihypertension, analgesic, anti-inflammatory, anti-Alzheimer, antipsychotic and/or anticoagulant agents.
Conclusions
This review article sheds a light on the most recent studies proving the importance of piperidine nucleus in the field of drug discovery.
Background
Heterocyclic compounds, Mainly those bearing Nitrogen are ubiquitous in nature and in medicaments as well. Most of them play important roles in the human body and have great biologic qualities.
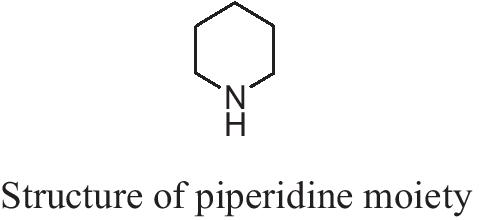
Piperidine (Hexahydropyridine) (Fig. 1) is an organic heterocyclic amine widely used as a building block and reagent in synthesizing organic compounds, Including medicinal products [1].
Piperidine derivatives represent a key and extensive category of Nitrogen bearing heterocyclic compounds. Compounds with piperidine moiety show a wide variety of biologic activities. Piperidine is a vital fundament in the production of drugs. This nitrogen bearing heterocyclic ring system and its byproducts show several essential features and are being utilized in different ways as anticancer, antimicrobial, analgesic, antiinflammatory, and antipsychotic agents. Piperidine and pyridine complexes comprehend two of the most common heterocyclic fragments present in FDA approved drugs [2].
Main text
Naturally occurring piperidine-based compounds
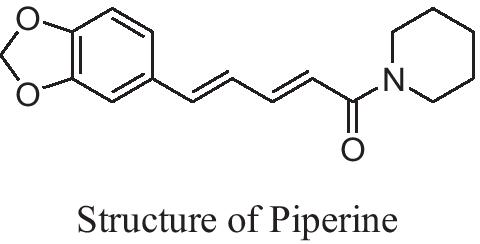
Piperine (N-acylpiperidine) (Fig. 2) is a true alkaloid having piperidine moiety, found in plants of Piperaceae family; it shows powerful antioxidant action because of its capability of hindering or suppressing free radicals. The pharmaceutical worth of Piperine is extensive because of its activity against cancer, inflammation, hypertension, and asthma [3]. Piperine was also found to have antidepressant like effects when given to mice with chronic mild stress [4]. On the other hand, the mechanism of action of Piperine is still not totally clear [5].
Nobuji et al. obtained diverse phenolic amides from pepper; those compounds showed reassuring anti-oxidation qualities in different assays. All of the amides displayed a positive antioxidation effect [6].
Several piperidine alkaloids isolated from natural herbs, were found to exhibit antiproliferation and antimetastatic effects on various types of cancers both in vitro and in vivo for example Piperine (Fig. 2), Evodiamine (Fig. 8), Matrine (Fig. 3), Berberine (Fig. 4) and Tetrandine (Fig. 5) [7].
Piperidine derivatives as anticancer agents
Cancer, uncontrolled cell growth, is a worldwide health matter that impacts a major proportion of the human population. These three malignant properties of cancer differentiate them from benign tumors, which are self-limited, do not invade or metastasize [8]. Cancer treatments have developed through recent years. Cancer treatments now include surgery, chemotherapy, radiotherapy, and modern approaches, namely interventional radiology, hormone therapy, and immunotherapy [9]. Anticancer medicines are categorized into groups according to their mechanism of action into alkylating agents, antimetabolites, anti-tumor antibiotics, topoisomerase inhibitors, and microtubule binding agents [10]. There are more than one hundred medically approved chemotherapeutic medications [11]. However, the toxicological data of most anticancer drugs have restrained their medicinal application as antiproliferative agents [10, 12]. In spite of the great achievements made in chemotherapy in recent years, resistance to conventional chemotherapeutic drugs and new targeted therapeutic agents is still a significant issue in cancer treatments and the cause of most recurrences [12,13,14,15,16]. Therefore, research of new anticancer agents with new mechanisms of action, high potency and low side effects is essential for chemists [8]. To diminish anticancer drugs' adverse effects upon normal cells, targeted cancer therapies are favored nowadays [17]. Targeted drugs block the growth and proliferation of cancer by interfering with specific molecules that are involved in the rise, advancement, and expansion of cancer without affecting normal cells [17]. Some chemotherapeutic agents have piperidine moiety within their structure, foremost among them, vinblastine and raloxifene.
Vinblastine (Fig. 6) is a plant alkaloid which is extracted from Vinca Rosea. This drug is an antineoplastic agent. It is thought that the mechanism of action of Vinblastine is inhibition of mitosis at metaphase leading to mitotic arrest or cell death [18].
Raloxifene (Fig. 7) is second-generation selective estrogen receptor modulator (SERM). It is used for reducing the risk of invasive breast carcinoma in postmenopausal women by acting as estrogen blocker [19].
Evodiamine (Fig. 8) which is a quinolone alkaloid isolated from Evodia rutaecarpa was found to show anticancer effects in vivo and in vitro by induction of apoptosis or cell cycle arrest therefore preventing metastasis and angiogenesis [20,21,22].
Khairia et al. synthesized two piperidine analogues. These analogues were evaluated for their chemo preventive effect. Compound 1 (Fig. 9) was found to be the most potent chemo-preventive agent and could successfully decrease the number of cancer cells [23].
Suvankar et al. studied a series of twenty-five piperidine derivatives as free radical scavengers. These compounds exhibited a potent anticancer effect. Compound 2 (Fig. 10) was found to be the most potent antitumor agent. The mechanism of action was found to be binding to ctDNA via intercalation [24].
Amitabh et al. synthesized a novel series of piperidine derivatives and related aryl methyl ethers that were evaluated against MCF-7 breast cancer cell line and were found to have potent cytotoxic activity. Compounds 3a and 3b (Fig. 11) showed the highest efficiency. These compounds were tested against murine L1210, human HeLa, and CEM cell lines and demonstrated greater activity than both tamoxifen and raloxifene [25].
Yanqun et al. designed and synthesized a string of piperidine derivatives as new HSP70 inhibitors based on virtual library screening. Among these, compounds 4a–4e (Fig. 12) showed remarkable inhibitory effect on human breast cancer cells [26].
Jin et al. synthesized a series of N-(piperidine-4-yl) benzamide compounds and investigated their effect against cancer cells. Structure activity relationship showed that the presence of halogen, carboxyl, nitro, or methyl groups on ring B increased the cytotoxicity of the Piperidine derivatives. Compound 5 (Fig. 13) was found to have the maximum potency with an IC50 value 15-fold higher than Sorafenib [27].
Dong et al. prepared a series of new piperidine derivatives. These analogues were assessed for their anticancer activity. Compound 6 (Fig. 14) expressed strong antiproliferative activity by inhibiting the tubulin polymerization [28].
Xin et al. prepared a line of innovative 5-phenyl-N-piperidine ethanone-4,5-dihydropyrazole by products. The novel compounds were evaluated for their anticancer activity. Among these, compound 7 (Fig. 15) exhibited maximum potency against SGC-7901and MGC-803 cell lines. Structure activity relationship studies showed that Introduction of amide or nitro functional groups in place of trifluoromethyl group led to decreased anticancer activity [29].
Benaka et al. prepared a series of new diphenyl (piperidine-4-yl) methanol derivatives. These compounds were evaluated for their antiproliferative activity against HT-29, HeLa,16 MCF-7 and HepG2 cell lines via MTT assay. Compound 8 (Fig. 16) showed high cytotoxic activity against all cell lines. Structure activity relationship studies exhibited that increasing the carbon chain length reduced the antitumor activity, presence of hydrophobic groups (Cl, F) are essential for binding with the active site of the enzyme, and presence of electron donating groups (Cl, F) promotes the antitumor effect [30].
Debasish et al. synthesized a string of eight pyrenyl derivatives that were also screened for antiproliferative action. According to the IC50 data, compounds 9a and 9b (Fig. 17) were the most potent cytotoxic agents in the string. According to structure activity relationship analysis, using four carbon linker produced higher cytotoxic effect than five carbon linker[31].
Jin et al. developed a series of novel P2X7 receptor antagonists bearing piperidine moiety as possible antitumor agents. Among them, compounds 10, 11 and 12 (Fig. 18) demonstrated highest cytotoxic effect against MDA-MB-231 breast cancer cell lines [32].
Piperidine derivatives with antiviral activity
Nikolai et al. synthesized a string of (R) and (S) isomers of novel adamantane-substituted piperidine analogs. Biologic tests of the produced compounds were carried out on the rimantadine-resistant S31N mutated strains of influenza A 2009(H1N1)pdm09 and modern pandemic strain 2016(H1N1)pdm09. The most powerful compounds were both enantiomers of the enol ester 13 (Fig. 19) exhibiting IC value equals to 7.7 µM with the 2016 Orenburg strain [33].
Guoxin et al. performed modifications on the structure of an original compound appointed from high throughput screening. A series of different analogues were prepared and were proved to have an inhibitory effect against influenza virus. Structure–activity relationship studies indicated that the ether linkage between quinoline and piperidine is crucial to the inhibitory effect. Compound 14 (Fig. 20) showed a superb inhibitory effect on a diversity of influenza virus strains and showed a good ability to interfere with the early to middle stages of influenza virus replication[34].
Piperidine derivatives with antimicrobial activity
AD et al. produced two piperidine derivatives. These derivatives were isolated from Streptomyces ficellus then evaluated for their antibacterial activity. Nojirimycin (Fig. 21) was found to be effective against S. lutea, S. aureus and E. coli. Nojirimycin is the prototype of a new class of antibiotics [35].
Chennan et al. prepared a string of piperidine-4-one oxime derivatives that was investigated for their in vitro antimicrobial activity. Compounds 15a and 15b (Fig. 22) exhibited strong antifungal activity against Aspergillus flavus and Candida-51, respectively. Compound 15b was found to be more effective than the reference drug; amphotericin B regarding MIC [36].
Jayaraman et al. synthesized a series of piperidone derivatives via Mannich reaction pathways. The compounds were evaluated for their antimicrobial effect against various types of bacteria and fungi. Compound 16a and 16b (Fig. 23) showed good antibacterial activity against B. subtilis, E. coli, K. pneumoniae and S. faecalis. Compound 17 and 16b (Fig. 23) showed good antifungal activity against A. niger, A flavus, C. neoformans, Candida 6 and Candida 51 [37].
Piperidine derivatives with antimalarial activity
Rokhyatou et al. designed and prepared novel 1,4 disubstituted piperidine derivatives that were tested for their activity against chloroquine sensitive and chloroquine resistant strains of P. falciparum. Compounds 18a–c (Fig. 24) were the most potent compounds among the prepared library [38].
Piperidine derivatives with antifungal activity
Zhigan et al. designed and synthesized a string of new antifungal piperidinyl triazole derivatives. Multiple compounds exhibited superb antifungal activity against a range of significant fungi. Among these compounds, compounds 19 and 20 (Fig. 25) were noticed to be highly active antifungal drugs [39].
Piperdine derivatives with antihypertension activity
Shamim et al. synthesized a string of novel N-substituted phenacyl piperidine derivatives that have been tested for their hypotensive activity. Compounds 21, 22a and 22b (Fig. 26) exhibited mild antihypertension activity. It was also discovered that the carboxamide group has no significant effects on arterial blood pressure and has no significant effect on blood pressure [40].
Yanjiang et al. tested compounds that were determined through screening of BMP2 up-regulators for their capacity to boost the expression of Inhibitor of DNA binding 1 (ID1) proteins. Depending on stem cell technology, they produced compound 23 (Fig. 27) as BMP upregulator that regulates BMP2 and PTGS2 levels, which may be worthwhile for treating pulmonary arterial hypertension [41].
SK et al. synthesized a novel piperidinyl urea derivatives that were tested for their activity as epoxide hydrolase inhibitors. Compound 24 (Fig. 28) was found to reduce the blood pressure effictively [42].
Piperidine derivatives with antiAlzheimer's activity
Gunhild et al. studied the effect of Donepezil (Fig. 29) on the development of apathy and other neuropsychiatric disorders in patients with Alzheimer’s disease. It was proved that Donepezil could cause a great reduction of apathy that reached over 6 months and thus had a considerable favourable effect [43].
Khairia et al. designed and synthesized a string of novel piperidine derivatives that were evaluated for their inhibitory effect on acetylcholinesterase. These derivatives exhibited successful AchE inhibition, high selectivity and significant radical scavenging properties. Compound 25 (Fig. 30) exhibited high free radical scavenging effect [44].
Piperidine derivatives with analgesic and antiinflammatory activity
Adel prepared a series of piperidine derivatives that were tested for their antitumour activity against most of the human tumour cells. All the assessed compounds exposed significant antitumour properties against (HCT-116), (T-47D), [HL-60 (TB), MOLT-4, RPMI-8226] and (PC-3) cell lines. Of all the assessed compounds, compounds 26a–c (Fig. 31) showed substantial antiinflammatory properties [45].
Ahmed et al. synthesized a line of 2,4,6-trisubstituted-quinazoline derivatives bearing piperidine moiety. These compounds were tested for analgesic and antiinflammatory activity. Of all the compounds analyzed, compounds 27b, 27c and 27d (Fig. 32) were found to have higher analgesic effect than the standard drug indomethacin. Compounds 27a, 27b, 27c, 27e, 28 and 29 (Fig. 32) displayed both analgesic and antiinflammatory effects [46].
Piperidine derivatives with antipsychotic activity
Takeo et al. studied and investigated the antipsychotic effect of a novel piperidine derivative. Compound 30 (Fig. 33) was observed to have affinity to several dopamine receptors and exhibited atypical antipsychotic effect in animal models with limited prevelance of extrapyramidal adverse effects [47].
Ling et al. synthesized a series of new benzoxazole-piperidine derivatives with high affinity to dopamine D2, serotonin 5-HT1A and serotonin 5HT2A receptors. Among these derivatives, compound 31 (Fig. 34) was found to be a promising antipsychotic agent [48].
Xudong et al. designed and synthesised a line of piperidine derivatives. Among them, Compound 32 (Fig. 35) showed special pharmacological properties and a worthwhile pharmacokinetic profile and exhibited pro-cognition properties. Compound 32 is thought to represent a new class of atypical antipsychotics for schizophrenia [49].
Piperidine derivatives with anticoagulant activity
Modesto et al. synthesized a line of 4-(piperidin-1-yl)pyridine derivatives. Structure activity relationship studies revealed that compound 33 (Fig. 36) exhibited strong factor IIa inhibition and showed good anticoagulant effect [50].
Akiyoshi et al. designed, prepared and investigated two racemic piperidine diamine derivatives. Compounds 34a–e (Fig. 37) expressed great inhibitory effect on factor Xa and displayed a great anticoagulant activity [51].
Khairia et al. designed and prepared a series of carbamoylpyridine and carbamoylpiperidine derivatives. These derivatives were investigated for their platelet aggregation inhibition effect. Among them, compounds 35 and 36 (Fig. 38) were identified to be the most potent antiplatelet aggregation agents [52].
Conclusions
This review mentions the potentiality of piperidine moiety accompanied with other molecular fragments in ameliorating various disease conditions. Naturally occurring piperidines were able to display extensive effects against cancer, inflammation, hypertension and sometimes behave as antioxidants. On the other hand, Synthetic piperidine derivatives that were presented in many research studies proved their potentiality as anticancer agents, antiviral, antimicrobial, antimalarial and antifungal agents. Also, piperidine derivatives can be designed and synthesized to exhibit additional biological effects as antihypertension, antiAlzheimer's, analgesic, anti-inflammatory, antipsychotic and anticoagulant activity.
Availability of data and material
Data and material are available upon request.
Abbreviations
- FDA:
-
Food and Drug Administration
- SERM:
-
Second-generation selective estrogen receptor modulator
- ctDNA:
-
Cell-free circulating tumor DNA
- MCF-7:
-
Type of breast cancer cell lines
- murine L1210:
-
Type of leukemia cancer cell lines
- human HeLa:
-
Type of cervical cancer cell lines
- CEM:
-
Type of leukemia cancer cell lines
- HSP70:
-
Heat Shock Protein Coding gene.
- SGC-7901:
-
Type of human gastric cancer cell lines
- MGC-803:
-
Type of human gastric cancer cell lines
- HT-29:
-
Type of human colorectal adeno cancer cell lines
- HepG2:
-
Type of human liver cancer cell lines
- MTT:
-
3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyl-2H-tetrazolium bromide assay
- P2X7:
-
Purinergic receptor
- MDA-MB-231:
-
Type of human breast adeno cancer cell lines
- BMP:
-
Basic Metabolic Panel
- PTGS:
-
Post-transcriptional gene silencing
- AchE:
-
Acetylcholine Esterase
- MOLT-4:
-
Type of leukemia cancer cell lines
- RPMI-8226:
-
Human myeloma cell line
- PC-3:
-
Human prostate cancer cell line
References
World Heritage Encyclopedia Piperidine. In: World Heritage Encyclopedia. http://self.gutenberg.org/articles/Piperidine. Accessed 10 Dec 2020
Vitaku E, Smith DT, Njardarson JT (2014) Analysis of the structural diversity, substitution patterns, and frequency of nitrogen heterocycles among U.S. FDA approved pharmaceuticals. J Med Chem 57:10257–10274
Manjusha RK, Begum S, Begum A, Bharathi K (2018) Antioxidant potential of piperidine containing compounds: a short review. Asian J Pharm Clin Res 11:66–73
Li S, Wang C, Wang M, Li W, Matsumoto K, Tang Y (2007) Antidepressant like effects of piperine in chronic mild stress treated mice and its possible mechanisms. Life Sci 80:1373–1381
de Almeida GC, Oliveira LFS, Predes D, Fokoue HH, Kuster RM, Oliveira FL, Mendes FA, Abreu JG (2020) Piperine suppresses the Wnt/β-catenin pathway and has anti-cancer effects on colorectal cancer cells. Sci Rep 10:1–12
Nakatani N, Inatani R, Ohta H, Nishioka A (1986) Chemical constituents of peppers (Piper spp.) and application to food preservation: naturally occurring antioxidative compounds. Environ Health Perspect 67:135–142
Lu JJ, Bao JL, Chen XP, Huang M, Wang YT (2012) Alkaloids isolated from natural herbs as the anticancer agents. Evid Based Complement Altern Med 2012:1–12
Singhal S, Ramanathan K, Shanthi V, Rajasekaran R (2012) Development of cancer database by amalgamation of various existing database. Int J Pharm Pharm Sci 4:479–483
Mayo Clinic Staff (MayoClinic) Cancer treatment. https://www.mayoclinic.org/tests-procedures/cancer-treatment/about/pac-20393344. Accessed 15 Dec 2020
The American Cancer society medical and editorial content team. https://www.cancer.org/treatment/treatments-and-side-effects/treatment-types/chemotherapy/how-chemotherapy-drugs-work.html#written_by. Accessed 20 Dec 2020
Arroyo M, Berral-Gonzalez A, Bueno-Fortes S, Alonso-Lopez DDLRJ (2020) Mining drug-target associations in cancer: analysis of gene expression and drug activity correlations. Biomolecules 10:667
Alfarouk KO, Stock CM, Taylor S, Walsh M, Muddathir AK, Verduzco D, Bashir AHH (2015) Resistance to cancer chemotherapy: failure in drug response from ADME to P-gp. Cancer Cell Int 15:71
Holohan C, Van Schaeybroeck S, Longley DBJP (2013) Cancer drug resistance: an evolving paradigm. Nat Rev Cancer 13:714–726
Rueff JRA (2016) Cancer drug resistance: a brief overview from a genetic viewpoint. Methods Mol Biol 1395:1–18
Borst P (2012) Cancer drug pan-resistance: pumps, cancer stem cells, quiescence, epithelial to mesenchymal transition, blocked cell death pathways, persisters or what? Open Biol 2:120066–120066
Housman G, Byler S, Heerboth S, Lapinska K, Longacre M, Snyder NSS (2014) Drug resistance in cancer: an overview. Cancers (Basel) 6:1769–1792
Mailankody S, Prasad V (2015) Five years of cancer drug approvals: innovation, efficacy, and costs. JAMA Oncol 1(4):539–540
Ngan VK, Bellman K, Hill BT, Wilson L, Jordan MA (2001) Mechanism of mitotic block and inhibition of cell proliferation by the semisynthetic vinca alkaloids vinorelbine and its newer derivative vinflunine. Mol Pharmacol 60:225–232
Taurin S, Allen KM, Scandlyn MJ, Rosengren RJ (2013) Raloxifene reduces triple-negative breast cancer tumor growth and decreases EGFR expression. Int J Oncol 43:785–792
Ogasawara M, Matsubara T, Takahashi S, Saiki I, Suzuki H (2002) Anti-invasive and metastatic activities of evodi-amine. Biol Pharm Bull 25:1491–1493
Fei XF, Wang BX, Li TJ, Tashiro S, Minami M, Xing DJ, Ikejima T (2003) Evodiamine, a constituent of Evodiae Fructus, induces anti-proliferating effects in tumor cells. Cancer Sci 94(1):92–98
Zhang Y, Wu LJ, Tashiro S, Onodera S, Ikejima T (2003) Intracellular regulation of evodiamine-induced A375–S2 cell death. Biol Pharm Bull 26(11):1543–1547
Youssef KM, Ezzo AM, El-Sayed MI, Hazzaa AA, EL-Medany AH, Arafa M, (2015) Chemopreventive effects of curcumin analogs in DMH-induced colon cancer in albino rats model. Future J Pharm Sci 1:57–72
Das S, da Silva CJ, Silva MM, Dantas MDA, de Fátima Â, Góis Ruiz ALT, da Silva CM, de Carvalho JE, Santos JCC, Figueiredo IM, da Silva-Júnior EF, de Aquino TM, de Araújo-Júnior JX, Brahmachari G, Modolo LV (2018) Highly functionalized piperidines: free radical scavenging, anticancer activity, DNA interaction and correlation with biological activity. J Adv Res 9:51–61
Jha A, Yadav Y, Naidu AB, Rao VK, Kumar A, Parmar VS, MacDonald WJ, Too CK, Balzarini J, Barden CJ, Cameron TS (2015) Design, synthesis and bioevaluation of novel 6-(4-Hydroxypiperidino)naphthalen-2-ol-based potential selective estrogen receptor modulators for breast cancer. Eur J Med Chem 92:103–114
Zeng Y, Cao R, Zhang T, Li S, Zhong W (2015) Design and synthesis of piperidine derivatives as novel human heat shock protein 70 inhibitors for the treatment of drug-resistant tumors. Eur J Med Chem 97:19–31
Hou J, Zhao W, Huang ZN, Yang SM, Wang LJ, Jiang Y, Zhou ZS, Zheng MY, Jiang JL, Li SH, Li FN (2015) Evaluation of novel N-(piperidine-4-yl)benzamide derivatives as potential cell cycle inhibitors in HepG2 cells. Chem Biol Drug Des 86:223–231
Fu DJ, Liu SM, Yang JJ, Li J (2020) Novel piperidine derivatives as colchicine binding site inhibitors induce apoptosis and inhibit epithelial-mesenchymal transition against prostate cancer PC3 cells. J Enzyme Inhib Med Chem 35:1403–1413
Liu XH, Li J, Shi JB, Song BAQX (2012) Design and synthesis of novel 5-phenyl-N12 piperidine ethanone containing 4, 5-dihydropyrazole derivatives as potential 13 antitumor agents. Eur J Med Chem 51:294–299
Prasad SB, Vinaya K, Kumar CA, Swarup S, Rangappa KS (2009) Synthesis and in vitro antiproliferative activity of diphenyl (sulphonylpiperidin-4-yl) methanol derivatives. Med Chem Res 19:220–235
Bandyopadhyay D, Sanchez JL, Guerrero AM, Chang FM, Granados JC, Short JD, Banik BK (2015) Design, synthesis and biological evaluation of novel pyrenyl derivatives as anticancer agents. Eur J Med Chem 89:851–862
Park J, Williams DR, Lee J, Lee SD, Lee JH, Ko H, Lee GE, Kim S, Lee JM, Abdelrahman A, Muller CE, Jung DW, Kim YC (2016) Potent suppressive effects of 1-piperidinylimidazole based novel P2X7 receptor antagonists on cancer cell migration and invasion. J Med Chem 59(16):7410–7430
Kuznetsov NY, Tikhov RM, Godovikov IA, Medvedev MG, Lyssenko KA, Burtseva EI, Kirillova ES, Bubnov YN (2017) Stereoselective synthesis of novel adamantane derivatives with high potency against rimantadine-resistant influenza A virus strains. Org Biomol Chem 15(15):3152–3157
Wang G, Chen L, Xian T, Liang Y, Zhang X, Yang Z, Luo M (2014) Discovery and SAR study of piperidine-based derivatives as novel influenza virus inhibitors. Org Biomol Chem 12:8048–8060
Argoudelis AD, Reusser F, Mizsak SA, Baczynskyj L (1976) Antibiotics produced by streptomyces ficellus. II. Feldamycin and nojirimycin. J Antibiot (Tokyo) 29:1007–1014
Ramalingan C, Park YT, Kabilan S (2006) Synthesis, stereochemistry, and antimicrobial evaluation of substituted piperidin-4-one oxime ethers. Eur J Med Chem 41:683–696
Jayabharathi J, Manimekalai A, Vani TC, Padmavathy M (2007) Synthesis, stereochemistry and antimicrobial evaluation of t(3)-benzyl-r(2), c(6)-diarylpiperidin-4-one and its derivatives. Eur J Med Chem 42:593–605
Seck R, Gassama A, Cojean S, Cavé C (2020) Synthesis and antimalarial activity of 1,4-disubstituted piperidine derivatives. Molecules 25:21–26
Jiang Z, Gu J, Wang C, Wang S, Liu N, Jiang Y, Zhang W, Sheng C (2014) Design, synthesis and antifungal activity of novel triazole derivatives containing substituted 1,2,3-triazole-piperdine side chains. Eur J Med Chem 82:490–497
Zafar S, Akhtar S, Ali SI, Mushtaq N, Naeem S, Ali M (2019) Synthesis, characterization and antimicrobial activity of piperidine derivatives. J Chem Soc Pak 41:363–367
Xing Y, Zhao S, Wei Q, Gong S, Zhao X, Zhou F, Ai-Lamki R, Ortmann D, Du M, Pedersen R, Shang G, Si S, Morrell NW, Yang J (2018) A novel piperidine identified by stem cell-based screening attenuates pulmonary arterial hypertension by regulating BMP2 and PTGS2 levels. Eur Respir J 51(4):1702229
Anandan SK, Webb HK, Chen D, Wang YX, Aavula BR, Cases S, Cheng Y, Do ZN, Mehra U, Tran V, Vincelette J, Waszczuk J, White K, Wong KR, Zhang LN, Jones PD, Hammock BD, Patel DV, Whitcomb R, MacIntyre DE, Sabry J, Gless R (2011) 1-(1-acetyl-piperidin-4-yl)-3-adamantan-1-yl-urea (AR9281) as a potent, selective, and orally available soluble epoxide hydrolase inhibitor with efficacy in rodent models of hypertension and dysglycemia. Bioorg Med Chem Lett 21(3):983–988
Waldemar G, Gauthier S, Jones R, Wilkinson D, Cummings J, Lopez O, Zhang R, Xu Y, Sun Y, Knox S, Richardson S, Mackell J (2011) Effect of donepezil on emergence of apathy in mild to moderate Alzheimer’s disease. Int J Geriatr Psychiatry 26:150–157
Youssef KM, Fawzy IM, El-subbagh HI (2017) N-substituted-piperidines as novel anti-Alzheimer agents: synthesis, antioxidant activity, and molecular docking study. Future J Pharm Sci 4:1–7
Girgis AS (2009) Regioselective synthesis and stereochemical structure of anti-tumor active dispiro[3H-indole-3,2′-pyrrolidine-3′,3″-piperidine]-2(1H),4″-diones. Eur J Med Chem 44:1257–1264
Alafeefy AM, Kadi AA, Al-deeb OA, El-tahir KEH, Al-jaber NA (2010) Synthesis, analgesic and anti-inflammatory evaluation of some novel quinazoline derivatives. Eur J Med Chem 45:4947–4952
Funakoshi T, Chaki S, Kawashima N, Suzuki Y, Yoshikawa R, Kumagai T, Nakazato A, Kameo K, Goto M, Okuyama S (2002) In vitro and in vivo pharmacological profile of 5-[2-[4-(6-fluoro-1H-indole-3-yl)piperidin-1-yl]ethyl]-4-(4-fluorophenyl)thiazole-2-carboxylic acid amide (NRA0562), a novel and putative atypical antipsychotic. Life Sci 7(12):1371–1384
Huang L, Zhang W, Zhang X, Yin L, Chen B, Song J (2015) Synthesis and pharmacological evaluation of piperidine (piperazine)-substituted benzoxazole derivatives as multi-target antipsychotics. Bioorg Med Chem Lett 5(22):5299–5305
Cao X, Zhang Y, Chen Y, Qiu Y, Yu M, Xu X, Liu X, Liu BF, Zhang L, Zhang G (2018) Synthesis and biological evaluation of fused tricyclic heterocycle piperazine (piperidine) derivatives as potential multireceptor atypical antipsychotics. J Med Chem 61:10017–10039
de Candia M, Fiorella F, Lopopolo G, Carotti A, Romano MR, Lograno MD, Martel S, Carrupt PA, Belviso BD, Caliandro R, Altomare C (2013) Synthesis and biological evaluation of direct thrombin inhibitors bearing 4-(piperidin-1-yl) pyridine at the P1 position with potent anticoagulant activity. J Med Chem 56:8696–8711
Mochizuki A, Nakamoto Y, Naito H, Uoto K, Ohta T (2008) Design, synthesis, and biological activity of piperidine diamine derivatives as factor Xa inhibitor. Bioorg Med Chem Lett 18:782–787
Youssef KM, Al-Omar MA, El-subbagh HI, Abou-zeid LA (2011) Synthesis, antiplatelet aggregation activity, and molecular modeling study of novel substituted-piperazine analogues. Med Chem Res 20:898–911
Acknowledgements
Not applicable.
Funding
Not applicable.
Author information
Authors and Affiliations
Contributions
MMA collected the data and wrote the draft manuscript. IMF collected the data and wrote the draft manuscript. HIES revised the manuscript. KMY revised the manuscript and supervised the whole work. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Abdelshaheed, M.M., Fawzy, I.M., El-Subbagh, H.I. et al. Piperidine nucleus in the field of drug discovery. Futur J Pharm Sci 7, 188 (2021). https://doi.org/10.1186/s43094-021-00335-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s43094-021-00335-y