- Review
- Open access
- Published:
Versatile mechanisms of 2-substituted benzimidazoles in targeted cancer therapy
Future Journal of Pharmaceutical Sciences volume 6, Article number: 41 (2020)
Abstract
Background
The aim of this review is to provide an overview on diverse anticancer activities of 2-substituted benzimidazole derivatives.
Main body
This review provides a correlation between the various mechanisms of action of benzimidazoles as anticancer and the substitution pattern around the nucleus.
Conclusion
The linker group and substitution at N-1, C-2, C-5, and C-6 positions have been found to be the most contributory factors for anticancer activity. This will help in the further design to afford more selective, potent, and multi-target anticancer of 2-substituted benzimidazole-based compounds.
Background
Cancer, the uncontrolled, rapid, and pathological proliferations of abnormal cells, is one of the most formidable afflictions in the world jeopardizing human life and health [1,2,3]. It is the second most life-threatening disease after the cardiovascular disorder according to statistics from the American Cancer Society (ACS) [4, 5]. Cancer can occur in any living cell at any stage of life; the morbidity and mortality associated with disease is on rise. Millions of people worldwide are suffering from this dreaded disease which not only affects the health of the patient but also puts significant socioeconomic, mental, and physical burden on the family members [6,7,8].
Heterocyclic compounds occupy a central position in medicinal chemistry and are of particular interest and significant importance in the search for new bioactive molecules in the pharmaceutical industry [9]. More interest in the area of benzimidazole-based chemistry was developed in the 1950s, when 5,6-dimethyl-1-(α-d-ribofuranosyl) benzimidazole was found as an integral part of the structure of vitamin B12 [10]. Benzimidazole being an isostere of purine-based nucleic acid found to be a privileged lead nucleus widely used in the design of many biologically active molecules [11]. Benzimidazoles exhibit a wide range of biological activities including antibacterial [12], antifungal [13], analgesic [14], and cardiovascular [15] in addition to anticancer activities [16,17,18].
Main text
Chemistry of benzimidazoles
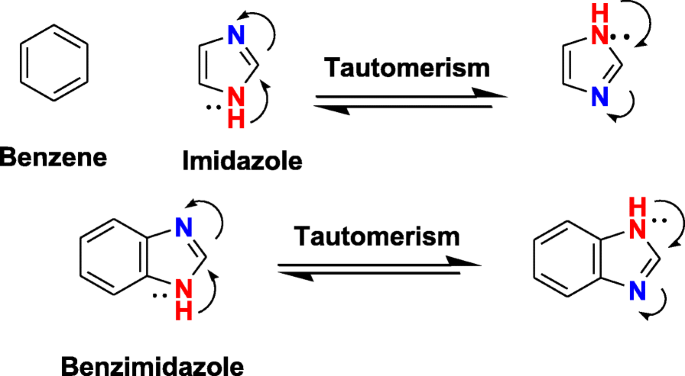
Benzimidazole is one of the oldest known nitrogen heterocycles and was first synthesized by Hoebrecker [19] then later by Ladenburg [20]. Benzimidazole ring contains two nitrogen atoms with amphoteric nature, possessing both acidic and basic characteristics (Fig. 1). It exists in two equivalent tautomeric forms, in which the hydrogen atom can be located on either of the two nitrogen atoms [16, 18].
Synthesis of 2-substituted benzimidazoles
The synthetic strategy adopted for the preparation of benzimidazoles relies on building up the imidazole ring on a suitable substituted benzene core.
The first benzimidazole was prepared by Hoebrecker [19]. who obtained 2,5-dimethylbenzimidazole by the reduction and dehydration of 2-nitro-4-methylacetanilide (Scheme 1) [21, 22].
In the majority of cases, o-phenylenediamines or suitably substituted derivative was used as the starting compound where it was reacted with different one carbon delivering reagents to give the benzimidazole derivatives (Scheme 2). The following reactions were classified according to the reagent used through different pathways from A to R [21,22,23,24].
Anticancer activity of 2-substituted benzimidazoles
Recently, the major side effect associated with the traditional anticancer agents is cytotoxicity towards normal cells due to lack of selectivity for the abnormal cells is well noticeable. With the rapid development of cell biology and molecular biology, the strategy of anti-cancer drug research has shifted to new and more selective targets for cancer cell proliferation, such as cancer cell signal transduction pathways, growth factors and their receptors, and apoptosis pathway. Therefore, the search for anticancer agents has been in continuum for many years.
Nevertheless, targeted therapy has some limitations; the chief among them is the potential for the cells to develop resistance. Unfortunately, in most cases, another targeted therapy that could overcome this resistance is not available, so it is advantageous to use targeted therapy in combination, either with other targeted therapy or with traditional therapy [25, 26].
In this review, a literature survey on the anticancer activity of benzimidazole was illustrated. Various anticancer agents (also referred to as antitumor, antiproliferative, and antineoplastics) reported for treatment of varied kinds of cancers act through different mechanisms. Benzimidazole exerted their antitumor activity through versatile mechanisms of action such as DNA alkylation, DNA binding, disturbing tubulin polymerization or depolymerization, enzyme inhibition, antiangiogeneic, and signal transduction inhibitors.
2-Substituted benzimidazoles as alkylating agents
Alkylating agents are highly electrophilic compounds that react with nucleophilic groups on DNA, in particular the N-7 of G base, to form a strong covalent bond disrupting replication or transcription.
1,2,5-Trisubstituted benzimidazoles were found to act as alkylating agents, for example, bendamustine 1 (TREANDA®), which has been approved by the FDA for the treatment of leukemia, multiple myeloma, and non-Hodgkin’s lymphoma [27, 28].
In addition, the pyrrolo[1,2-a]benzimidazoles-4,7-diones 2a–c are one of the early classes of anticancer agents designed as new DNA cross-linkers acting through cleavages of G and A bases which possess IC50 ranging from 10 to 6000 nM values against various human ovarian and colon cancer cell lines [29].

Benzimidazoles as DNA-binding agents
This class of compounds is known broadly as antimetabolites. These drugs inhibit the enzymes involved in the synthesis of nucleotide building blocks of DNA, resulting in the inhibition of DNA function which may lead to apoptosis [11].
Gao et al. synthesized 2-substiuted novel benzimidazole acridine subsidiaries keeping in setting that acridines can intercalate into DNA and benzimidazoles can tie in the DNA minor groove. MTT assay showed that most of the designed synthesized compounds revealed great antiproliferative action, among which compound 3 exhibited high potency against both chronic myelogenous leukemia K562 and liver HepG2 cells [30].
2,6-Disubstituted benzimidazole-oxindole conjugate derivatives were synthesized by Nayak et al. furthermore, investigated the apoptotic system initiated by these conjugates against human breast cancer cell line, MCF-7. Following evaluation, compounds 4a and 4b showed 43.7% and 43.6% and 64.8% and 62.7% apoptosis, respectively, at 1 and 2 μM concentrations [31].
On the other hand, the 5-substituted bisbenzimidazoles Hoechst 33258 (5a) and Hoechst 33342 (5b) were designed as DNA minor groove–binding agents. They displayed in vitro antitumor activities and nonspecifically inhibit the catalytic proliferation of DNA enzymes [32].
Derivatives of 1,2,5-trisubstitued 1-(4-methoxyphenethyl)-1H-benzimidazole-5-carboxylic acid were prepared and assessed as potential chemotherapeutic agents by Gowda et al. Among synthesized analogs methyl 1-(4-methoxyphenethyl)-2-(4-fluoro-3-nitrophenyl)-1H-benzimidazole-5-carboxylate 6 provoked cell death in leukemic cells with an IC50 of 3 μM. Additionally, it stimulates S/G2 cell cycle arrest, and downregulation of cyclin B1, CDK2, and PCNA. Moreover, the replacement of fluorine and nitro with amine, chlorine, or bromine reduced the activity as shown by the SAR study [33].
Hranjec et al. designed and synthesized benzimidazo[1,2-a]quinolones candidates that have the capacity to be incorporated into the space between the DNA base pairs resulting in DNA cleavage. All compounds exerted pronounced antiproliferative activity on five tumor cell lines, whereby compound 7 exerted the highest activity on all cell lines (IC50 = 0.7–25 μM) and showed a special selectivity towards HeLa cells [34].
Recently, more 1,2-fused planar benzimidazole derivatives have been reported to exhibit potent cytotoxicity, for example, (pyrimidobenzimidazolone) 8 with in vitro NSCLC growth % from − 2.92 to 38.33 and GI50 ranges from − 6.13 to − 4.82 μM. Compound 9 (1,3-diarylpyrazino benzimidazole) exhibited in vitro % inhibition of lipid peroxidation (AAPH); interaction % with DPPH (2,2-diphenyl-1-picrylhydrazyl)and in vitro % inhibition of soybean lipoxygenase (LOX % inh) was found to be 84, 5, 19 %, respectively [35, 36].
Zhou et al. developed and synthesized a new Cu(II) complex of benzimidazoles 10 possessed antitumor activity. The results suggested that the complex could electrostatically bind to the phosphate group of DNA backbone and partially intercalate into the double helix of DNA because of the bulky structure of the complex and the planarity of the benzimidazole rings [37].

2-Substituted benzimidazoles as tubulin inhibitors
Microtubules are the key components of the cytoskeleton of eukaryotic cells composed of α/β-tubulin heterodimers. They are involved in intracellular migration and transport. Tubulin inhibitors thus act by interfering with the dynamics of microtubules, i.e., growing (polymerization) and shortening (depolymerization). Either way, it leads to mitotic arrest and cell death [38,39,40].
Lu et al. outlined the synthesis and assessment of a series of tubulin inhibitors. Structure-activity relationships of these compounds prompted recognizable proof of new 2,4-disubstituted benzimidazole and imidazo(4,5-c)pyridine-fused ring templates, represented by compounds 11 and 12, respectively. Compound 11 enhanced the metabolic stability in liver microsomes and was the most active of the series with IC50 0.022 ± 0.009 μM and 0.025 ± 0.004 against prostate tumor cell PC-3 and melanoma cancer cell A375, respectively [41].
Kamal et al. identified a series of 2,6-disubstituted pyrazolyl benzimidazole analogs as microtubule-destabilizing agents where they showed potent growth-inhibitory activity against most of the sixty cancer cell line panels of NCI ranging from 0.3 to 13 μM. A549 cells treated with 13a–c-arrested cells at the G2/M phase apart from activating cyclin-B1 protein levels and disrupting the microtubule network. Moreover, these conjugates adequately hindered tubulin polymerization with IC50 values of 1.3–3.8 μM. Compound 13b with a methoxy substituent showed obvious cytotoxic potential and incited activity right around 3-fold higher than CA-4 [42].
2,5-Disubstituted novel benzimidazole carbamates linked with indole moieties by sulfur or selenium atoms were synthesized and examined for their inhibitory action against some human cancer cell lines. Among these, compound 14 was identified as the most active antitumor against HT-1080, A-549, and SGC-7901 human cancer cell lines with IC50 values ranging from 0.098 to 0.15 μM. In vitro tubulin polymerization assay revealed that 14 significantly detains tubulin polymerization and upsets tubulin microtubule. Also, a molecular modeling study exhibited that compound 14 bonds with a coupling mode similar to that of nocodazole [43].
Analogs of 2,5-disubstituted benzimidazoles were developed and assessed for their tubulin inhibitory activity. Compound 15 strongly restrained the proliferation of a panel human cancer cells, with IC50 values ranging from 0.006 to 1.774 μM; also it prompted cell cycle arrest at the G2/M phase [44].
2,4/5-Disubstituted class of terphenyl benzimidazoles 16a,b were designed and synthesized as tubulin polymerization inhibitors. 16a,b demonstrated significant anticancer activity with GI50 ranges from < 0.1 to 2.11 μM. These compounds exhibit G2/M phase arrest; besides the analysis of tubulin by western blot, experiments were carried out revealing a concomitant decrease in the polymerized tubulin [45].
Wang et al. designed some 1,2-disubstituted benzimidazole derivatives as potential tubulin polymerization inhibitors and anthropic cancer cell line cytotoxic agents. Compound 17 was established as the most potent tubulin polymerization inhibitory agent with IC50 of 1.5 μM and exhibited antiproliferative activity against several human cancer cell lines [46].

2-Substituted benzimidazoles as enzyme inhibitors
2-Substituted benzimidazoles as sirtuin inhibitors
Sirtuins are class III histone deacetylases (HDACs) that catalyze the deacetylation of proteins. They focus on an extensive variety of cell proteins in the nucleus, cytoplasm, and mitochondria [47, 48]. It has been reported that SIRT1 and SIRT2 are upregulated in numerous tumor sorts and they are able to inactivate some tumor suppressor proteins [49]. Therefore, the development of novel sirtuin inhibitors has been proposed as another line in the treatment of cancer [50, 51].
Yoon et al. developed novel 1,2,5-trisubstituted benzimidazole derivatives as SIRT1 and SIRT2 inhibitors. Compound 18 showed the best inhibitory activity for SIRT1 (IC50 = 54.21 μM) and for SIRT2 (IC50 = 26.85 μM). Cell proliferation assay demonstrated that compound 18 had pronounced antitumor activity against three different types of cancer cells (breast MDA-MB-468, colon HCT-116, and blood-leukemia CCRF-CEM). The SAR study verified that the replacement of the phenyl group at position N1 led to a loss of activity [52].

2-Substituted benzimidazoles as poly (ADP-ribose) polymerase inhibitors
Poly (ADP-ribose) polymerase-1 (PARP-1) is a key DNA damage–sensing enzyme that facilitates the repair of DNA. PARP-1 contributes to the resistance that often develops after cancer therapy. Consequently, it is assumed that in vivo inhibition of PARP will block DNA repair and increase the maximum therapeutic benefit of chemotherapy [53].
Some 2,4-disubstituted benzimidazoles were demonstrated to have PARP inhibitory activity. Penning et al. reported the design of a new series of 2-substituted-1H-benzimidazole-4-carboxamides. Especially, compound 19 Veliparib (ABT-888) displayed great adequacy against both the PARP-1 and PARP-2 enzymes with a Ki (inhibitory constant and reflective of the binding affinity) of 5 nM and in a C41 whole-cell assay with an EC50 of 2 nM and exhibited potentiation of two cytotoxic agents in particular temozolomide and cisplatin in both mouse melanoma and breast cancer demonstrate [54].
Abdullah et al. prepared different 2,4,6-trisubstituted benzimidazole derivatives and studied their activity as dual Poly (ADP-ribose) polymerases (PARPs) and Dihydroorotate dehydrogenase (DHODH) inhibitors. Analogs 20a-d were proven to be the most potent compounds of the series with IC50 ranges from 0.013–55 to 0.061–44 μM against DHODH and PARPs, respectively [55].
A series of novel 2,4,5-trisubstituted benzimidazole analogs were identified and evaluated for their PARP-1 inhibitory activity by Wang et al. All target compounds demonstrated high intrinsic PARP-1 inhibitory activity and have been assessed for in vitro cellular assays to evaluate the potentiation effect of cytotoxic agents against cancer cell lines. Compound 21 showed strong inhibition against the PARP-1 enzyme with an IC50 of 43.7 nM, excellent cell inhibitory activity in HCT116 cells (IC50 = 7.4 μM), and potentiation of temozolomide cytotoxicity in the cancer cell line A549 (PF50 = 1.6) [56].
Zhou et al. synthesized several derivatives of 2-substituted-3,4-fused benzimidazole carboxamide as PARP-1 inhibitors, which can be 18F-labeled easily for positron emission tomographic (PET) imaging. Of the compounds synthesized, 22 had the highest inhibition potency for PARP-1 with IC50 6.3 nM [57].

2-Substituted benzimidazoles as methionine synthase inhibitors (MetSIs)
Enzyme methionine synthase catalyzes the transfer of a methyl group from 5-methyltetrahydrofolate to homocysteine, to obtain methionine and tetrahydrofolate; thus, inhibition of methionine synthase (MetS) would settle DNA and RNA [58].
One of the most potent inhibitors of MetS was 2,5-disubstituted benzimidazole 23 which showed IC50 of 50 ± 5 μM with a score of the lowest free energy of binding − 1610.42 kJ/mol [59].
Other 2,5-disubstituted benzimidazole derivatives were designed and evaluated for their inhibitory activity against purified rat liver methionine synthase using a radiometric enzyme assay. Compounds 24a,b were proven to be the most powerful compounds, with IC50 values of 20 μM and 18 μM, respectively. Modeling and docking studies determine the molecular requirements of the activity of this class of compounds [60].

2-Substituted benzimidazoles as DHFR inhibitors
Dihydrofolate reductase (DHFR) is a critical enzyme in folate metabolism. It converts dihydrofolate (DHF) to tetrahydrofolate (THF), which is essential for purine and thymidylate (TMP) synthesis in cell proliferation. Depressing DHFR activity results in THF deficiency and disruption of subsequent DNA replication and resulting in cell death.
A new series of 1,2,6-trisubstituted triazine-benzimidazole hybrid was developed as a potent DHFRI. Compound 25 was found to be the most active DHFRI with IC50 of 1.05 μM. These findings are the outcome of their inhibitory activities tested over 60 human tumor cell lines, whereas 25 revealed a broad spectrum of antitumor activities with a GI50 value of 9.79 μM [11, 61].
Recently, Singla et al designed and synthesized novel series of 1,2,6-trisubstituted quinazolin-4-one-benzimidazoles. As per the results of dihydrofolate reductase enzyme immunoassay, compound 26 exhibited comparable activity with IC50 value of 0.011 μM in contrast to methotrexate (IC50 = 0.02 μM). The preparatory interactive examinations with calf thymus DNA by UV–visible and fluorescence spectroscopy disclosed that compound 26 was viably intercalated with ct-DNA [62].

2-Substituted benzimidazoles as topoisomerase I and II inhibitors
Topoisomerase enzymes are a family of enzymes which catalyze the cleavage, annealing, and supercoiling of DNA and considered a major target for antineoplastic agents used in the treatment of breast, lung, and prostate cancer, sarcomas, and hematological malignancies. All topoisomerase II-directed agents are able to interfere with at least one step of the catalytic cycle [63, 64].
2-Substituted benzimidazole derivatives 27a,b were synthesized and screened against topoisomerase I with inhibition percent 95.4%, 90.2%, respectively. Meanwhile, the cytotoxic activity against HeLa (cervix adenocarcinoma), MCF7 (breast adenocarcinoma), and A431 (skin epidermoid carcinoma) cells were measured, the IC50 were found to range from 6.04 to > 30 μM [65].
A series of new pyrazolo[3,4-d]pyrimidine possessing 4-(1H-benzimidazol-2-yl)-phenylamine moiety has been designed and synthesized by single et al. Compounds 28, 29, 30, and 31 turned out to be the most efficacious candidates in this series, with mean GI50 values of 1.30 μM, 1.43 μM, 2.38 μM, and 2.18 μM, respectively, against several cancer cell lines. Moreover, these compounds induced apoptosis and suppressed human topoisomerase (Topo) IIα. These discoveries established a framework for the sound outline of powerful agents for probing proteins, which are relied upon to give vital knowledge into the field of DNA and protein connections [66].
In a study of some novel fused heterocyclic compounds as eukaryotic topisomerase II inhibitors. It was discovered that 5-methylcarboxylate-2-phenylthiomethylbenzimidazole, 32, with IC50 17 μM was more active than the reference drug etoposide ( IC50 = 21.8 μM) [67].
Singh et al. worked on the synthesis of a novel class of topoisomerase I inhibitor as 2-aryl-5-substituted-2,5-bisbenzimidazole derivatives. Analogs 33a,b were found to be with the highest ability to induce DNA cleavage with IC50 in the micromolar range (0.6–5.5) against U87, MCF7, and HeLa human tumor cells. The SAR study revealed that the introduction of halogen groups at the phenyl ring increased the binding affinity to ct-DNA particularly fluoro and chloro groups which showed significant cytotoxic activity to human tumor cell lines [68].
Jin et al. prepared a series of heterocyclic derivatives of 5-phenyl-bibenzimidazoles, 34, and assessed them for their topoisomerase I poisoning activity and cytotoxicity. It was concluded that topoisomerase I poisoning activity was related to analogs that had a hydrogen atom capable of hydrogen bond formation, thus influencing the activity [69].
A new Cu2+ complex of 2-pyridinylbenzimidazole-5-carboxylic acid analogs were designed and prepared by Galal et al. Among all, compound 35 was found to be the most potent candidate inhibiting the topoisomerase II on the DNA-relaxing activity of P388 topoisomerase II with IC50 2.5 μM. Meanwhile, the growth-inhibitory effects of the tested compounds on 21 human solid tumor cell lines (8 lung, 7 colon, and 6 gastric cancer cell lines) with mean GI50 = 0.091 μM [70].

2-Substituted benzimidazoles as androgen receptor activity antagonists
Testosterone and dihydrotestosterone control protein anabolism and influence basal metabolism through the upregulation of the androgen receptor. Although androgens have numerous valuable impacts and are critical for sexual orientation and male advancement, endogenous androgens, for example, testosterone fortify hyperplasia of the prostate and intensify androgen-subordinate prostate disease [71].
Elancheran et al. designed and synthesized a series of 1,3,5-tri-substituted 2-oxobenzimidazoles derivatives and investigated them as androgen receptor antagonistic activity. It can be concluded that compound 36 is the most active compound from the series against prostatic PC-3 and LNCaP cancer cell lines [72].
Ng et al. have investigated some 1,2,5,6-tetrasubstituted benzimidazoles as androgen receptor antagonists for their utilization in prostate malignancy. The SAR studies have prompted the 1-(4-bromobenzyl) derivative 37 as the most intense androgen receptor opponent which has an ID50 of 0.13 mg/day [73].

2-Substituted benzimidazoles as α-glucosidase inhibitors
α-Glucosidase inhibitors have wide biological significance as chemotherapeutic agents for the treatment of carbohydrate mediated diseases such as diabetes and cancer [74].
Consequently, 2,5,6-trisubstituted benzimidazoles bearing thiourea moiety have been synthesized and assessed for α-glucosidase inhibition using Baker’s yeast α-glucosidase enzyme. All tested compounds exhibited variable α-glucosidase inhibitory activity, while compound 38 showed significant inhibitory effects with an IC50 value of 35.83 ± 0.66 μM, which was more potent than the standard acarbose (IC50 = 774.5 ± 1.94 μM) [75].

2-Substituted benzimidazoles as the G9a histone methyltransferase inhibitors
G9a is initially identified as a H3K9 methyltransferase that specifically mono- and dimethylates “Lys-9” of histone H3 (H3K9me1 and H3K9me2, respectively) in euchromatin. Histone lysine methylation plays a central epigenetic role in the organization of chromatin domains and the regulation of gene expression [76]. Mutation and amplification of HMTs are frequently observed in human cancers and employed as a promising target in cancer therapy [77].
Recently, 1,2,5-trisubstituted benzimidazole scaffold was discovered as G9a histone methyltransferase inhibitor by Zhang et al. Based on structure optimization, 24 structural analogs are designed and synthesized. The kinase inhibition assay showed that compound 39 potently inhibits G9a with an IC50 of 1.32 μM. Besides, the MTT assay revealed that MCF-7 is most sensitive to 39 among five different breast cancer cells with IC50 5.73 ± 0.95. In addition, compound 39 induces obvious autophagy in MCF7 cells by fluorescence microscope assays and western blot analysis [78].

2-Substituted benzimidazoles as pyruvate kinase inhibitors
Recently, the role of the M2 isoform of pyruvate kinase in the change in cellular metabolism to aerobic glycolysis has been proposed [79]. Tumor cells entirely express the embryonic M2 isoform of pyruvate kinase (PKM2); the dimeric form assists cell growth by increasing glycolytic intermediates for biosynthetic processes, but when energy levels decrease, the enzyme shifts to the tetrameric form and makes oxidative phosphorylation easy [80].
Consequently, the discovery of 1,2-disubstituted benzimidazole series was described as potent and selective PKM2 activators by Guo et al.; Compound 40 was reported to have appreciable inhibitory activity against PKM2 cancer cells with a lactate dehydrogenase (LDH) IC50 value of 3.5 μM [81].

2-Substituted bisbenzimidazoles as telomerazse inhibitors
Maji et al. reported a new series of 2,6-disubstituted bisbenzimidazole-carbazoles and was evaluated for their antiproliferative activity as telomerase inhibitors where compound 41 was recognized as the most potent derivative with an IC50 value of 0.6 ± 0.01 μM [82].
Similarly, 2-substituted tribenzimidazole derivatives were synthesized and evaluated for their telomerase inhibitory properties. Compound 42 was found to be the most active compound of the series [83].
In addition, compounds 43a-c are derivatives of 2-substituted-5,6-fused benzimiadzole that exhibited in vitro inhibition of hTERT expression, telomerase inhibition, and suppression of prostatic cancer cell growth with IC50 values 5.1 to 27.9 μM [84].

2-Substituted benzimidazoles as antiangiogenic agents and signal transduction inhibitors
Tumor growth and development of distant metastasis are supported through angiogenesis where new blood vessels extend from pre-existing ones. This process is mediated via the vascular endothelial growth factor (VEGF) which links to its receptor tyrosine kinase (RTK) to advance the proliferation and survival of endothelial cells. Consequently, using kinase inhibitors to adjust VEGFR signaling is considered as a powerful method for the inhibition of tumor angiogenesis [85, 86].
Protein kinases regulate most aspects of normal cellular function, especially signals transduction from the cell membrane into the interior of the cell [87]. Signal transduction relates to the methods by which regulatory molecules that control essential processes of cell growth and differentiation, convey within the cell. Most malignancies have abnormal or overexpressed signal transduction factors which depend on the kinase enzymes thus recommending them as targets for therapeutic progress [88, 89].
A large number of benzimidazoles were reported to possess protein kinases inhibitory activity [90].
2-Substituted benzimidazoles as VEGFR inhibitors
Keeping this in mind, 2-substituted benzimidazole analogs were prepared and assessed for their in vivo antitumor and antiangiogenic activities. Compounds 44 and 45 showed cytotoxic effects on MDA-MB-31 cells and HUVEC with IC50 value ranges from 0.40 to 6 μM and from 0.05 to 1.5, respectively, and effectively antagonized VEGF-A165/NRP-1 binding [91].
In addition, a series of 2,5/6-disubstituted benzimidazole derivatives were developed as potent VEGFR-2 (KDR) kinase inhibitors. Among them, compound 46 showed the most potent VEGFR-2 inhibitory activity with an IC50 value of 0.03 μM and it also demonstrated strong anticancer activity against the tested cancer cell lines [85].
Temirak et al. identified 1-aryl,2-furyl benzimidazoles and tested their anti-cancer activity where compound 47 showed the most potential antiangiogenic effect against VEGFR2 kinase with an IC50 value of 6.98 μg/mL [92].
A novel series of 1,2-disubstituted benzimidazol-furan hybrids were designed, synthesized, and evaluated for their in vitro cytotoxic activity against breast (MCF-7) and hepatocellular (HepG2) carcinoma cell lines 48a,b, 49-51. Two of the synthesized conjugates, 48b and 49, showed potent antiproliferative properties against the MCF-7 cell line (IC50 = 21.25 and 21.35 μM, respectively). Additionally, compounds 48a,b, 50, and 51 showed promising potency (IC50 = 25.95, 22.58, 26.94, and 31.06 μM, respectively) against the liver carcinoma cell line HepG2. Meanwhile, the in vitro evaluation on VEGFR-2 in the MCF-7 cell line showed their potent inhibitory activity ranges from 92–96%, compound 48a was found to have promising VEGFR-2 inhibitory activity (IC50 = 0.64 μM) [93].
Novel conjugates of 2,5,6-trisubstituted benzimidazoles were synthesized and examined against VEGFR-1 and VEGFR-2. Compound 52 displayed VEGFR-2 inhibitory activity with a 50% inhibition concentration value as low as 0.02 ± 0.03 μM. VEGFR-2 active compounds display good activity against VEGFR-1 up to 91% inhibition at 10 μM concentration. The compounds likewise give a chance of establishing the framework for promising molecules of anticancer activity [94].

2-Substituted benzimidazoles as EGFR inhibitors
The EGFR (also known as erbB1 or HER1) and the related human epidermal growth factor receptor HER2 (also known as erbB2) is an encouraging aim for anticancer drug design because of its value in tumor growth, metastasis, and angiogenesis [95,96,97,98].
Akhtar et al. identified the synthesis of oxadiazole attached to benzimidazole at position C2 as potential EGFR and erbB2 receptor inhibitors and assessed their cytotoxic activity. The most active compounds against breast cancer cell lines constitute para-substituted chloro/methoxy phenyl at the fifth position of oxadiazole 53a,b, while, derivative 53a specifically affected cell cycle arrest at G2/M phase with an increase in apoptosis. Also, it inhibited EGFR and erbB2 receptor at 0.081 and 0.098 μM concentration [99]. A series of 1,2,7-trisubstituted benzimidazole derivatives were developed and evaluated for mutant non-small-cell lung cancer activity and epidermal growth factor receptor (EGFR) by Lelais et al. Compound 54 was proven to be the most powerful compound of the series against lung cancer cell lines [100].

2-Substituted benzimidazoles as FGFR1 inhibitors
Fibroblast growth factor receptor (FGFR) represents an attractive oncology target for cancer therapy in perspective of its basic part in advancing tumor formation and progression, and additionally making resistance-affirmed treatments [101].
Yan et al. synthesized a series of 2,5-disubstituted benzimidazole and evaluated them for effective inhibition effect of the fibroblast growth factor receptor. Compound 55 was recognized as a potent pan-FGFR inhibitor and exhibited outstanding in vitro inhibitory activity against a panel of FGFR-amplified cell lines with IC50 < 0.1 nM. Also, 55 gave almost total control of tumor development (96.9 % TGI) in NCI-H1581 (FGFR1-amplified) xenograft mice model [102].
New 2,6-disubstituted benzmidazoles were synthesized by Gryshchenko et al. and assessed for inhibition of FGFR1 kinase activity. Compounds 56a,b displayed the most potent activity with an IC50 value of 0.63 μM and 0.32 μM respectively. The SAR study showed that the presence of the hydroxyl group at the meta position of the benzene ring of 56a,b caused rapid augmentation of inhibition towards FGFR1 [103].

2-Substituted benzimidazole Tie-2 receptor tyrosine kinase inhibitors
Tie-2 is a receptor tyrosine kinase that is essential for the formation of the embryonic vasculature and is strongly implicated in tumor angiogenesis [104].
Novel 2,5-disubstituted benzimidazolyl-4-aminopyrrolopyrimidine analogs 57a,b were identified as potent inhibitors of the Tie-2 kinase. Compound 57a with 2,6-di-fluoro-phenylsulfonyl group was discovered to have Tie-2 kinase and cellular (Caco-2BA/AB) IC50 of 568 and 518 nM, respectively. The corresponding urea analog compound 57b was more potent inhibitor against Tie-2 kinase cellular assays with IC50 of 241 and 112 nM, respectively [87].

2-Substituted benzimidazoles as heparanase inhibitors
Heparanase is an endo β-d-glucuronidase that cleaves heparan sulfate polymers in the extracellular matrix and regulates the release of many growth factors that are involved in tumor invasion. The finding that heparanase is elevated in a variety of tumors and is subsequently linked to the development of pathological processes makes the inhibition of this enzyme a target for anticancer development programs [105].
Pan et al. incorporated a 2,5,6-trisubstituted bisbenzimdazole with urea derivatives and assessed their potential to suppress heparanase enzyme. Compound 58 revealed high heparanase inhibitory activity with IC50 ranging from 0.075–0.27 μM [106].

2-Substituted benzimidazoles as COX-1 and COX-2 inhibitors
In the aim of developing novel technique for cancer treatment, cyclooxygenase enzymes inhibitors was introduced [107]. Cyclooxygenase proteins (COX) are the principle organizers of prostaglandin (PG) biosynthesis from arachidonic acid. COX enzymes are classified principally into two isoforms: a constitutive form (COX-1) and an inducible form (COX-2). The COX-2 isoform was found to be excessively expressed in tough human malignancies, for example, breast cancer, bladder, prostate, and colon. The COX-2 enzyme plays a part in apoptosis control, by increasing angiogenesis [108].
In this context, new series of 2-substituted benzimidazole pyrazoles were prepared and evaluated for their antiproliferative activity against breast carcinoma (MCF-7) and non-small cell lung cancer (A549) cell lines. Compound 59 was the most active against both A549 and MCF-7 cell lines with IC50 = 8.46 and 6.42 μM, respectively. Also, 59 is the most COX-2 selective compound among all synthesized derivatives; it showed inhibitory activity against COX-2 enzymes with IC50 = 0.10 Μm, S.I. = 104.67) compared with celecoxib (COX-2 IC50 = 1.11 μM, S.I. = 13.33) [109].

2-Substituted benzimidazoles as Aurora A/B kinase inhibitors
Aurora kinases are serine-threonine kinases that play a critical role in the mitotic events of cell division [110]. They are essential to secure the correct progress of cell cycle during mitosis or meiosis [111]. Different benzimidazoles have been developed as Aurora kinase inhibitors for cancer treatment.
Novel 2,6-disubstituted benzimidazole derivatives have been designed and synthesized as Aurora kinase inhibitors. The entire target compounds were determined against cancer cell lines U937, K562, A549, LoVo, and HT29 and were screened for Aurora A/B kinase inhibitory activity in vitro. Ccompound 60 demonstrated selective cancer cell line inhibitory activity towards U937, LoVo, and HT29 with IC50 values 3.821, 1.422, and 1.299 μM, respectively. In addition, 60 revealed high Aurora A/B kinase inhibitory activities (IC50 124.9 and 191.0 nM) [112].
Compound 61, a 2-aminobenzimidazole derivative was identified and proven to act as potent Aurora kinase inhibitor with IC50 against Aurora A/B 17 and 5 nM, respectively. 2-Aminobenzimidazole acts as a bioisostere of the biaryl urea moiety of SNS-314, a potent Aurora kinase inhibitor, thus entered into the clinical study. This series of compounds present more aqueous solubility while keeping comparable potency in in vitro assays; compared with SNS-314, 61, in particular, has also exhibited a comparable profile to SNS-314 [113].
A novel 2,5-disubstituted benzimidazole (AT-9283) was described by Howard et al. as a multi-targeted kinase inhibitor, with special activity towards Aurora kinases A and B. Compound 62 (AT-9283) inhibited HCT116 cell line (IC50 = 0.03 μM) and demostrated the polyploid cellular phenotype characteristically related to Aurora B kinase inhibition (IC50 of approximately 3 nM). Compound 62 demonstrated in vivo efficacy in mouse xenograft models and is currently under evaluation in phase I clinical trials [114].
In an effort to identify novel compounds targeting Aurora kinase enzyme, Sharma et al. developed 1,2,5-trisubstituted benzimidazole derivatives. Among the several prepared compounds, 63 proved to be 1.25-fold more active than the positive control 5-FU, with a GI50 value of 18.12 μM (MG-MID). Moreover, interaction of the compound 48 with Aurora A enzyme showed selective inhibition of Aurora A kinase with IC50 value of 0.0 l μM [115].

2-Substituted benzimidazoles as checkpoint kinase (Chk1 and Chk2) inhibitors
Checkpoint kinase is a serine-threonine protein kinase that coordinates the DNA damage response and is activated by phosphorylation prompting cellular response such as cell cycle regulation, DNA repair, or apoptosis. ChK proteins are invariably more abundant in tumors as compared with normal tissues [116]. Inhibition of Chk2 has been proposed to be a significant argument of current cancer therapies [117, 118].
A series of 2/2,5-disubstituted benzimidazole derivatives was designed and synthesized by Song et al. as inhibitors of checkpoint kinase 1 (Chk1). Most of them exhibited moderate to good Chk1 inhibitory activities. Among them, compounds 64a–c showed significant Chk1 inhibitory activities with IC50 values of 4.05, 6.23, and 2.33 nM, respectively [119].
Some 2-substituted benzimidazoles were reported by Ni et al. as a novel class of small molecule ChK-1 inhibitors. Compound 65 has emerged as a potent and selective compound with IC50 value 0.32 nM [120].
Neff et al. have celebrated a new series of 2,5-disubstituted benzimidazole compounds containing pendant alcohol and amine moieties were found to be active against Chk2. Compound 66 was found to have the best inhibitory activity with IC50 value 14 ± 8 nM [121].
Galal et al. have applied a structure-based design to synthesize a new series of 2,5-di substituted benzimidazoles as a highly active and selective class of Chk2. The activities of the conjugates as checkpoint kinase inhibitors and as an antitumor were evaluated. The results indicated that compounds 67 and 68 inhibited Chk2 activity with high potency IC50 ranges from 5.5 to 52.8 nM [122].
A series of compounds comprising 2,5-disubstituted benzimidazole and dimethylpyrazolyl were synthesized. The cytotoxic activity of all compounds was tested against 60 types of human cancer cell lines. The results declared that compound 69 was found to be the most potent molecule against lung and breast cancers [123].

2-Substituted benzimidazoles as protein kinase rhCK2α inhibitors
Protein kinase 2 (CK2) is a constitutively active serine/threonine protein kinase which takes part in a direction of substantial varieties of processes identified with cell survival and multiplication including cell cycle, apoptosis, or angiogenesis [124]. CK2 is a generally appropriated enzyme that phosphorylates various regulatory proteins [125]. CK2 articulation appears to be overexpressed in numerous solid tumors; consequently, inhibition of CK2 activity can reduce the viability of cancer cells [126].
Schneider et al. described the synthesis and CK2 inhibitory activity of 2-substituted-4,5,6,7-tetra halogenated benzimidazole derivatives. It was observed that, compound 70 displayed the highest activity towards CK2 with a half maximal lethal dose (LD50) of 4.75 ± 1.02 μM [127].
Pagano et al. designed and synthesized conjugates of 2-substituted tetrabromo-benzimidazole derivatives and examined against CK2 enzyme activity. Upon evaluation, compound 71 was shown to display the lowest Ki value as a CK2 inhibitor (40 nM) [128].
In the same context, Andrzejewska et al. prepared 2-substituted polyhalogenobenzimidazoles and assessed the combined derivatives for their CK2 inhibitory activity utilizing CK2 pure from rodent liver cytosol. The most effective hindrance of CK2 was achieved just if the benzene ring of benzimidazole is tetrahalogenated which is obviously confirmed by the Ki value of 4,5,6,7-tetrabromo-2-trifluoromethyl benzimidazole, 72 (Ki = 0.60 μM) [129].
In addition, 1,2-disubstituted-1-H-benzimidazole was developed by Chojnacka et al. and assessed for protein kinase rhCK2α catalytic subunit inhibition and cytotoxicity against two cancer cell lines. Compound 73 was identified as the most active with a Ki value 2.42 ± 0.32 μM [130].

2-Substituted benzimidazoles as cyclin-dependant kinase inhibitors
Cyclin-dependent kinases (CDKs) constitute a class of serine-threonine protein kinases that play an important role in the regulation of the cell cycle [131].
Lin et al. reported the CDK inhibitory activity of a novel series of 2,4-disubstituted benzimidazolyl-pyrazolo[3,4-b]pyridines. The representative analog 74 was found to be a potent inhibitor of CDK1 with IC50 0.0056 μM and a significant decline of the in vitro cellular proliferation in HeLa, HCT116, and A375 human tumor cell lines with IC50 0.015, 0.010, and 0.010 μM, respectively [132].

2-Substituted benzimidazoles as insulin-like growth factor receptor-1 inhibitors
The insulin-like growth factor receptor-1 (IGF-1R) is a membrane receptor tyrosine kinase. It plays an important role in mutagenesis and cell survival [133, 134]. Overexpression of IGF-1R and IGF-1 was demonstrated in a variety of tumors, including glioma, lung, ovary, breast, carcinomas, sarcomas, and melanoma [135].
Consequently, 2,5,7-trisubstituted benzimidazole (BMS-53692470) 75 was discovered as a novel small inhibitor of IGF-1R IC50 100 nM [136].

2-Substituted benzimidazoles as allosteric mitogen-activated protein kinase (MEK1) and phosphatidylinositol 3-kinase (PI3K) inhibitors
The phosphoinositide 3-kinase (PI3K) pathway is a key signal transduction system that links oncogenes and multiple receptor classes to many essential cellular functions and is perhaps the most commonly activated signaling pathway in human cancer. There are four highly homologous isoforms, assigned PI3Kα, PI3Kβ, PI3Kγ, and PI3Kδ, each having an unmistakable cluster of physiological capacities [137]. Initiating changes in PI3Kα have been found in about a fourth of breast and endometrial tumors, recognizing PI3K as an imperative focus for novel growth therapeutics [138].
Recently, Dort et al. synthesized 1,2-disubstituted benzimidazole derivatives and displayed them as dual MEK/PI3K inhibitory agents by direct coupling of a potent PI3K inhibitor to an allosteric MEK inhibitor using a covalent linker moiety. A prototype dual-acting agent, compound 76 exhibited high in vitro inhibition of both PI3K (IC50 = 172 nM) and MEK1 (IC50 = 473 nM) [139].
A new series of 1,2-disubstituted benzimidazole pyrimidone derivatives were blended by Certal et al. and assessed them for treatment of phosphatase and TENsin homolog (PTEN)-deficient cancers as PI3Kβ has emerged as the isoform involved in the tumorigenicity of PTEN-deficient tumors. Among all compounds, 77, showed significant activity and selectivity for PI3Kβ (IC50 100 nM), achieved tumor growth delay with IC50 ranging from 76 to 188 nM and adequate in vitro pharmacokinetic properties. The SAR study showed that N-methyl benzimidazole compound resulted in a slight improvement of activity, compared with 5-fluoro derivatives [140].

2-Substituted benzimidazoles as a farnesyl-binding pocket of PDEδ inhibitors
K-Ras is one of the most common mutated signal-transducing human oncogenes. Ras signaling activity requires correct cellular localization of the GTPase. The spatial association of K-Ras is controlled by the prenyl-binding protein PDEδ that has a fundamental part in keeping up with the best possible cell dispersion of Ras proteins. Thus inhibition of the Ras-PDEδ cooperation by small molecules hinders Ras signaling [141].
Zimmermann et al. described the design of a novel 1,2-disubstituted benzimidazole derivatives as potent PDEδ supressors. Among the compounds developed, compound 78 was reported as the most active compound of the series against the farnesyl-binding pocket of PDEδ with KD 87 ± 35 nanomolar affinity as the introduction of a piperidine moiety acts as a hydrogen-bonding donor [142].
A series of 4-substituted derivatives of the pan class I PI 3-kinase inhibitor 2-(difluoromethyl)-1-[4,6-di-(4-morpholinyl)-1,3,5-triazin-2-yl]-1H-benzimidazole (ZSTK474) were prepared by Rewcastle et al. to synthesize a range of 1,2,4-trisubstituted benzimidazole derivatives with 4-aminoalkoxy substituents. The compounds were evaluated using two human tumor cell lines. Upon evaluation, it was found that compound 79 displayed the best enzyme inhibitor with IC50 ranges from 4.5 to 11 μM and revealed the highest antitumor activity with IC50 0.04 μM. Moreover, 79 showed the best overall activity against the U87MG xenograft model, but less potent than ZSTK474 [143].

2-Substituted benzimidazoles as dual c-Met and VEGFR-2 inhibitors
c-Met (mesenchymal endothelial transition) kinase is a subclass that is important, associated with its ligand, hepatocyte growth factor (HGF), for normal mammalian growth [144]. Targeting several biochemical paths of cancer can be accomplished by using several drugs with different modes of action or through a single moiety that could adjust multiple targets of a multi-component disorder [145].
Accordingly, 2,6-disubstituted benzoimidazolyl quinazolinamine derivatives were synthesized and evaluated by Shi et al. Compound 80 was found to be the most potent against c-Met and VEGFR-2 with of IC50 of 0.05 μM and 0.02 μM respectively [146].

2-Substituted benzimidazoles as multi-target receptor tyrosine kinase inhibitors
The development of resistance against drugs that act as a single kinase inhibitor makes inhibition of more than one protein kinase an acceptable idea because tumor cells are attacked concomitantly at several relevant targets. Moreover, if a single cancer-related kinase becomes drug-resistant, a multi-targeted drug would still act on the remaining array of target kinases. Some benzimidazoles with 2,6-disubstitution were found to have multi-target receptor tyrosine kinase inhibitory activity. Dovitinib, 81, a substituted 3-benzimidazol hydroquinolin-2-one, exhibited dual inhibition of FGFR (fibroblast growth factor receptor) and VEFGRs [147].
Li et al. designed a new series of 2,6-disubstituted benzimidazoles and evaluated their biological activity against HEPG-2 cells and different kinases (EGFR, PDGFR-a, PDGFR-b, VEGFR-2). Compound 82 was found to exhibit high cytotoxicity against HEPG-2 cells with an IC50 value of approximately 2 μM. Further kinase assay study showed that 82 has IC50 86.9, 18.7, and 16.5 μM against EGFR, VEFGR-2, and PDGFR, respectively [148].
McBride et al. synthesized 2,5-disubstituted benzimidazole indazole hybrids as multi-inhibitors of VEGFR-1, VEGFR-2, PDGFR, FGFR-1, and HUVEC and found that compounds 83 possessed favorable pharmacokinetics and exhibit impressive tumor growth inhibition property against different cell lines tested with IC50 0.028, 0.078, 0.69, 0.048, and 0.097 μM, respectively [149].
Determann et al. reported the synthesis of 1,2-disubstituted benzimidazole derivatives and evaluated against four cancer-related protein kinases namely Aurora B, PLK1 (polo-like kinase 1), FAK (focal adhesion kinase), and VEFGR-2, where compound 84 demonstrated high inhibitory activity with IC50 values 6.0 ± 0.2, 1.2 ± 0.2, 3.4 ± 0.8, 7.2 ± 0.3 μM, respectively [98].

Conclusion
This review highlights the current status of 2-substituted benzimidazole anticancer molecules. The linker group and substitution at N-1, C-2, C-5, and C-6 positions have been found to be the most contributory factors for anticancer activity. Table 1 of SM is a correlation between the type of substitution and the exerted mechanism of action. We hope this paper will form a comprehensive foundation and reference source that will open up new opportunities for researchers interested in drug designing of benzimidazoles as anticancer. This depends on the specific design of molecules targeting multiple receptors/enzyme/protein, particularly keeping in mind to lessen side effects and toxicity.
Availability of data and materials
Not applicable.
Abbreviations
- G:
-
Guanine
- A:
-
Adenosine
- IC50 :
-
Half maximal inhibitory concentration
- SAR:
-
Structure-activity relationship
- CDK2:
-
Cyclin-dependent kinase-2
- PCNA:
-
Proliferation cell nuclear antigen
- NCI:
-
National Cancer Institute
- NSCLC:
-
Non-small-cell lung cancer
- GI50 :
-
Half maximal inhibitory growth
- Ki:
-
Inhibitory constant and reflective of the binding affinity
- PF50 :
-
Potentiation effect
- HDAC:
-
Histone deacetylases
- PARP-1:
-
Poly (ADP-ribose) polymerase-1
- MetS:
-
Methionine synthase
- DHFR:
-
Dihydrofolate reductase
- ct-DNA:
-
Circulating tumor DNA
- ID50 :
-
Half maximal inhibitory dose
- HMTs:
-
Histone methyltransferase
- PKM2:
-
M2 isoform of pyruvate kinase
- hTERT:
-
Human telomerase reverse transcriptase
- VEGF:
-
Vascular endothelial growth factor
- RTK:
-
Receptor tyrosine kinase
- VEGFR:
-
Vascular endothelial growth factor receptor
- HUVEC:
-
Human umbilical vein endothelial cell
- KDR:
-
Kinase domain receptor
- EGFR:
-
Epidermal growth factor receptor
- FGFRs:
-
Fibroblast growth factor receptors
- TGI:
-
Tumor growth inhibition
- COX:
-
Cyclooxygenase
- S.I.:
-
Selective inhibition
- Chk:
-
Checkpoint kinase
- PK:
-
Protein kinase
- CDK:
-
Cyclin-dependent kinase
- PI3K:
-
Phosphoinositide 3-kinase
- MEK:
-
Mitogen-activated protein kinase
- PDEδ:
-
Prenyl-binding protein
- c-Met:
-
Cytoplasmic mesenchymal endothelial transition
- PDGFR:
-
Platelet-derived growth factor receptor
- GI:
-
Growth inhibition
- EC50 :
-
Median effective concentration required to induce a 50% effect
References
Nepali K, Sharma S, Sharma M, Bedi P, Dhar K (2014) Rational approaches, design strategies, structure activity relationship and mechanistic insights for anticancer hybrids. European Journal of Medicinal Chemistry 77:422–487
Fadeyi OO, Adamson ST, Myles EL, Okoro CO (2008) Novel fluorinated acridone derivatives. Part 1: Synthesis and evaluation as potential anticancer agents. Bioorganic & Medicinal Chemistry Letters 18(14):4172–4176
Rostom SA (2006) Synthesis and in vitro antitumor evaluation of some indeno [1, 2-c] pyrazol (in) es substituted with sulfonamide, sulfonylurea (-thiourea) pharmacophores, and some derived thiazole ring systems. Bioorganic & Medicinal Chemistry 14(19):6475–6485
Bajaj S, Asati V, Singh J, Roy PP (2015) 1, 3, 4-Oxadiazoles: an emerging scaffold to target growth factors, enzymes and kinases as anticancer agents. European Journal of Medicinal Chemistry 97:124–141
Ferlay J, Soerjomataram I, Dikshit R, Eser S, Mathers C, Rebelo M et al (2015) Cancer incidence and mortality worldwide: sources, methods and major patterns in GLOBOCAN 2012. International Journal of Cancer 136(5):E359–E386
Husain A, Rashid M, Mishra R, Parveen S, Shin D-S, Kumar D (2012) Benzimidazole bearing oxadiazole and triazolo-thiadiazoles nucleus: design and synthesis as anticancer agents. Bioorganic & Medicinal Chemistry Letters 22(17):5438–5444
Kidwai M, Venktaramanan R, Mohan R, Sapra P (2002) Cancer chemotherapy and heterocyclic compounds. Current Medicinal Chemistry 9(12):1209–1228
Lee CW, Hong DH, Han SB, Jung S-H, Kim HC, Fine RL et al (2002) A novel stereo-selective sulfonylurea, 1-[1-(4-aminobenzoyl)-2, 3-dihydro-1H-indol-6-sulfonyl]-4-phenyl-imidazolidin-2-one, has antitumor efficacy in in vitro and in vivo tumor models. Biochemical Pharmacology 64(3):473–480
Gaba M, Singh S, Mohan C (2014) Benzimidazole: an emerging scaffold for analgesic and anti-inflammatory agents. European Journal of Medicinal Chemistry 76:494–505
Toohey J, Barker H (1961) Isolation of coenzyme B12 from liver. Journal of Biological Chemistry 236(2):560–563
Akhtar J, Khan AA, Ali Z, Haider R, Shahar Yar M (2017) Structure-activity relationship (SAR) study and design strategies of nitrogen-containing heterocyclic moieties for their anticancer activities. European Journal of Medicinal Chemistry 125:143–189
Carcanague D, Shue Y-K, Wuonola MA, Uria-Nickelsen M, Joubran C, Abedi JK et al (2002) Novel Structures Derived from 2-[[(2-Pyridyl) methyl] thio]-1 H-benzimidazole as Anti-Helicobacter p ylori Agents, Part 2. Journal of Medicinal Chemistry 45(19):4300–4309
Aghatabay NM, Somer M, Senel M, Dulger B, Gucin F (2007) Raman, FT-IR, NMR spectroscopic data and antimicrobial activity of bis [μ 2-(benzimidazol-2-yl)-2-ethanethiolato-N, S, S-chloro-palladium (II)] dimer,[(μ 2-CH 2 CH 2 NHNCC 6 H 4) PdCl] 2· C 2 H 5 OH complex. European Journal of Medicinal Chemistry 42(8):1069–1075
Demirayak S, Karaburun AC, Kayagil I, Uçucu U, Beis R (2005) Synthesis and Analgesic Activities of Some 2-(Benzazolylacetyl) amino-3-ethoxycarbonylthiophene Derivatives. Phosphorus, Sulfur, and Silicon and the Related Elements 180(8):1841–1848
Locatelli A, Cosconati S, Micucci M, Leoni A, Marinelli L, Bedini A et al (2013) Ligand based approach to L-type calcium channel by imidazo [2, 1-b] thiazole-1, 4-dihydropyridines: from heart activity to brain affinity. Journal of Medicinal Chemistry 56(10):3866–3877
Yadav G, Ganguly S (2015) Structure activity relationship (SAR) study of benzimidazole scaffold for different biological activities: a mini-review. European Journal of Medicinal Chemistry 97:419–443
Gaba M, Gaba P, Uppal D, Dhingra N, Bahia MS, Silakari O et al (2015) Benzimidazole derivatives: search for GI-friendly anti-inflammatory analgesic agents. Acta Pharmaceutica Sinica B 5(4):337–342
Gaba M, Mohan C (2016) Development of drugs based on imidazole and benzimidazole bioactive heterocycles: recent advances and future directions. Medicinal Chemistry Research 25(2):173–210
Hobrecker F (1872) Reduction-products of nitracetamide compounds. Deut Chem Ges Ber 5:920–924
Ladenburg A (1879) Ueber einige Derivate der Tropasäure. Berichte der deutschen chemischen Gesellschaft 12(1):947–948
Wright JB (1951) The chemistry of the benzimidazoles. Chemical Reviews 48(3):397–541
Alaqeel SI (2017) Synthetic approaches to benzimidazoles from o-phenylenediamine: a literature review. Journal of Saudi Chemical Society 21(2):229–237
Zhu J, Zhang Z, Miao C, Liu W, Sun W (2017) Synthesis of benzimidazoles from o-phenylenediamines and DMF derivatives in the presence of PhSiH 3. Tetrahedron 73(25):3458–3462
Shrivastava N, Naim M, Alam M, Nawaz F, Ahmed S, Alam O (2017) Benzimidazole Scaffold as anticancer agent: synthetic approaches and structure–activity relationship. Archiv der Pharmazie.
Sawyers C (2004) Targeted cancer therapy. Nature 432(7015):294–297
Gerber DE (2008) Targeted therapies: a new generation of cancer treatments. American Family Physician 77(3):311–319
Lissitchkov T, Arnaudov G, Peytchev D, Merkle K (2006) Phase-I/II study to evaluate dose limiting toxicity, maximum tolerated dose, and tolerability of bendamustine HCl in pre-treated patients with B-chronic lymphocytic leukaemia (Binet stages B and C) requiring therapy. Journal of Cancer Research and Clinical Oncology 132(2):99–104
Husain A, Rashid M, Shaharyar M, Siddiqui AA, Mishra R (2013) Benzimidazole clubbed with triazolo-thiadiazoles and triazolo-thiadiazines: new anticancer agents. European Journal of Medicinal Chemistry 62:785–798
Schulz WG, Islam I, Skibo EB (1995) Pyrrolo [1, 2-a] benzimidazole-based quinones and iminoquinones. The role of the 3-substituent on cytotoxicity. Journal of Medicinal Chemistry 38(1):109–118
Gao C, Li B, Zhang B, Sun Q, Li L, Li X et al (2015) Synthesis and biological evaluation of benzimidazole acridine derivatives as potential DNA-binding and apoptosis-inducing agents. Bioorganic & Medicinal Chemistry 23(8):1800–1807
Nayak VL, Nagaseshadri B, Vishnuvardhan M, Kamal A (2016) Investigation of the apoptotic pathway induced by benzimidazole–oxindole conjugates against human breast cancer cells MCF-7. Bioorganic & Medicinal Chemistry Letters 26(14):3313–3317
Chen AY, Yu C, Bodley A, Peng LF, Liu LF (1993) A new mammalian DNA topoisomerase I poison Hoechst 33342: cytotoxicity and drug resistance in human cell cultures. Cancer Research 53(6):1332–1337
Gowda NT, Kavitha C, Chiruvella KK, Joy O, Rangappa KS, Raghavan SC (2009) Synthesis and biological evaluation of novel 1-(4-methoxyphenethyl)-1H-benzimidazole-5-carboxylic acid derivatives and their precursors as antileukemic agents. Bioorganic & Medicinal Chemistry Letters 19(16):4594–4600
Hranjec M, Pavlović G, Marjanović M, Kralj M, Karminski-Zamola G (2010) Benzimidazole derivatives related to 2, 3-acrylonitriles, benzimidazo [1, 2-a] quinolines and fluorenes: synthesis, antitumor evaluation in vitro and crystal structure determination. European Journal of Medicinal Chemistry 45(6):2405–2417
Neochoritis CG, Zarganes-Tzitzikas T, Tsoleridis CA, Stephanidou-Stephanatou J, Kontogiorgis CA, Hadjipavlou-Litina DJ et al (2011) One-pot microwave assisted synthesis under green chemistry conditions, antioxidant screening, and cytotoxicity assessments of benzimidazole Schiff bases and pyrimido [1, 2-a] benzimidazol-3 (4H)-ones. European Journal of Medicinal Chemistry 46(1):297–306
Demirayak S, Kayagil I, Yurttas L (2011) Microwave supported synthesis of some novel 1, 3-Diarylpyrazino [1, 2-a] benzimidazole derivatives and investigation of their anticancer activities. European Journal of Medicinal Chemistry 46(1):411–416
Zhou Q, Yang P (2006) Crystal structure and DNA-binding studies of a new Cu (II) complex involving benzimidazole. Inorganica Chimica Acta 359(4):1200–1206
Perez EA (2009) Microtubule inhibitors: differentiating tubulin-inhibiting agents based on mechanisms of action, clinical activity, and resistance. Molecular Cancer Therapeutics 8(8):2086–2095
Harris F, Pierpoint L (2012) Photodynamic therapy based on 5-aminolevulinic acid and its use as an antimicrobial agent. Medicinal Research Reviews 32(6):1292–1327
Jordan MA, Wilson L (2004) Microtubules as a target for anticancer drugs. Nature Reviews Cancer 4(4):253–265
Lu Y, Chen J, Wang J, Li C-M, Ahn S, Barrett CM et al (2014) Design, synthesis, and biological evaluation of stable colchicine binding site tubulin inhibitors as potential anticancer agents. Journal of Medicinal Chemistry 57(17):7355–7366
Kamal A, Shaik AB, Polepalli S, Kumar GB, Reddy VS, Mahesh R et al (2015) Synthesis of arylpyrazole linked benzimidazole conjugates as potential microtubule disruptors. Bioorganic & Medicinal Chemistry 23(5):1082–1095
Guan Q, Han C, Zuo D, Li Z, Zhang Q, Zhai Y et al (2014) Synthesis and evaluation of benzimidazole carbamates bearing indole moieties for antiproliferative and antitubulin activities. European Journal of Medicinal Chemistry 87:306–315
Wang W, Kong D, Cheng H, Tan L, Zhang Z, Zhuang X et al (2014) New benzimidazole-2-urea derivates as tubulin inhibitors. Bioorganic & Medicinal Chemistry Letters 24(17):4250–4253
Kamal A, Reddy MK, Shaik TB, Srikanth Y, Reddy VS, Kumar GB et al (2012) Synthesis of terphenyl benzimidazoles as tubulin polymerization inhibitors. European Journal of Medicinal Chemistry 50:9–17
Wang Y-T, Qin Y-J, Yang N, Zhang Y-L, Liu C-H, Zhu H-L (2015) Synthesis, biological evaluation, and molecular docking studies of novel 1-benzene acyl-2-(1-methylindol-3-yl)-benzimidazole derivatives as potential tubulin polymerization inhibitors. European Journal of Medicinal Chemistry 99:125–137
Blander G, Guarente L (2004) The Sir2 family of protein deacetylases. Annual Review of Biochemistry 73(1):417–435
Sebastián C, Satterstrom FK, Haigis MC, Mostoslavsky R (2012) From sirtuin biology to human diseases: an update. Journal of Biological Chemistry 287(51):42444–42452
Frye RA (2000) Phylogenetic classification of prokaryotic and eukaryotic Sir2-like proteins. Biochemical and Biophysical Research Communications 273(2):793–798
Saunders L, Verdin E (2007) Sirtuins: critical regulators at the crossroads between cancer and aging. Oncogene 26(37):5489–5504
Zhao W, Kruse J-P, Tang Y, Jung SY, Qin J, Gu W (2008) Negative regulation of the deacetylase SIRT1 by DBC1. Nature 451(7178):587–590
Yoon YK, Ali MA, Wei AC, Shirazi AN, Parang K, Choon TS (2014) Benzimidazoles as new scaffold of sirtuin inhibitors: green synthesis, in vitro studies, molecular docking analysis and evaluation of their anti-cancer properties. European Journal of Medicinal Chemistry 83:448–454
Drew Y, Mulligan EA, Vong W-T, Thomas HD, Kahn S, Kyle S et al (2011) Therapeutic potential of poly (ADP-ribose) polymerase inhibitor AG014699 in human cancers with mutated or methylated BRCA1 or BRCA2. Journal of the National Cancer Institute 103(4):334–346
Penning TD, Zhu G-D, Gandhi VB, Gong J, Liu X, Shi Y et al (2008) Discovery of the poly (ADP-ribose) polymerase (PARP) inhibitor 2-[(R)-2-methylpyrrolidin-2-yl]-1 H-benzimidazole-4-carboxamide (ABT-888) for the treatment of cancer. Journal of Medicinal Chemistry 52(2):514–523
Abdullah I, Chee CF, Lee Y-K, Thunuguntla SSR, Reddy KS, Nellore K et al (2015) Benzimidazole derivatives as potential dual inhibitors for PARP-1 and DHODH. Bioorganic & Medicinal Chemistry 23(15):4669–4680
Wang J, Wang X, Li H, Ji D, Li Y, Xu Y et al (2016) Design, synthesis and biological evaluation of novel 5-fluoro-1H-benzimidazole-4-carboxamide derivatives as potent PARP-1 inhibitors. Bioorganic & Medicinal Chemistry Letters 26(16):4127–4132
Zhou D, Chu W, Xu J, Jones LA, Peng X, Li S et al (2014) Synthesis,[18 F] radiolabeling, and evaluation of poly (ADP-ribose) polymerase-1 (PARP-1) inhibitors for in vivo imaging of PARP-1 using positron emission tomography. Bioorganic & Medicinal Chemistry 22(5):1700–1707
Zhou Z, Garrow TA, Dong X, Luchini DN, Loor JJ (2017) Hepatic activity and transcription of betaine-homocysteine methyltransferase, methionine synthase, and cystathionine synthase in periparturient dairy cows are altered to different extents by supply of methionine and choline. The Journal of Nutrition 147(1):11–19
Ahmed HA (2011) Molecular modeling and synthesis of certain substituted aryl compounds which have a potential anticancer activity. Bulletin of Faculty of Pharmacy, Cairo University 49(1):25–36
Elshihawy H, Helal MA, Said M, Hammad MA (2014) Design, synthesis, and enzyme kinetics of novel benzimidazole and quinoxaline derivatives as methionine synthase inhibitors. Bioorganic & Medicinal Chemistry 22(1):550–558
Singla P, Luxami V, Paul K (2015) Triazine–benzimidazole hybrids: anticancer activity, DNA interaction and dihydrofolate reductase inhibitors. Bioorganic & Medicinal Chemistry 23(8):1691–1700
Singla P, Luxami V, Paul K (2017) Quinazolinone-benzimidazole conjugates: synthesis, characterization, dihydrofolate reductase inhibition, DNA and protein binding properties. Journal of Photochemistry and Photobiology B: Biology 168:156–164
Larsen AK, Escargueil AE, Skladanowski A (2003) Catalytic topoisomerase II inhibitors in cancer therapy. Pharmacology & Therapeutics 99(2):167–181
Nitiss JL (2009) Targeting DNA topoisomerase II in cancer chemotherapy. Nature Reviews Cancer 9(5):338–350
Coban G, Zencir S, Zupkó I, Réthy B, Gunes HS, Topcu Z (2009) Synthesis and biological activity evaluation of 1H-benzimidazoles via mammalian DNA topoisomerase I and cytostaticity assays. European Journal of Medicinal Chemistry 44(5):2280–2285
Singla P, Luxami V, Singh R, Tandon V, Paul K (2017) Novel pyrazolo [3, 4-d] pyrimidine with 4-(1H-benzimidazol-2-yl)-phenylamine as broad spectrum anticancer agents: Synthesis, cell based assay, topoisomerase inhibition, DNA intercalation and bovine serum albumin studies. European Journal of Medicinal Chemistry 126:24–35
Pinar A, Yurdakul P, Yildiz I, Temiz-Arpaci O, Acan NL, Aki-Sener E et al (2004) Some fused heterocyclic compounds as eukaryotic topoisomerase II inhibitors. Biochemical and Biophysical Research Communications 317(2):670–674
Singh M, Tandon V (2011) Synthesis and biological activity of novel inhibitors of topoisomerase I: 2-Aryl-substituted 2-bis-1H-benzimidazoles. European Journal of Medicinal Chemistry 46(2):659–669
Jin S, Kim J, Sim S, Liu A, Pilch D, Liu F et al (2000) Design and structure–activity relationship of 3-benzimidazol-2-yl-1H-indazoles as inhibitors of receptor tyrosine kinases. Bioorganic and Medicinal Chemistry Letters 10:719–723
Galal SA, Hegab KH, Hashem AM, Youssef NS (2010) Synthesis and antitumor activity of novel benzimidazole-5-carboxylic acid derivatives and their transition metal complexes as topoisomerease II inhibitors. European Journal of Medicinal Chemistry 45(12):5685–5691
Kokontis JM, Liao S (1998) Molecular action of androgen in the normal and neoplastic prostate. Vitamins and Hormones 55:219–307
Elancheran R, Saravanan K, Choudhury B, Divakar S, Kabilan S, Ramanathan M et al (2016) Design and development of oxobenzimidazoles as novel androgen receptor antagonists. Medicinal Chemistry Research 25(4):539–552
Ng RA, Guan J, Alford VC, Lanter JC, Allan GF, Sbriscia T et al (2007) 2-(2, 2, 2-Trifluoroethyl)-5, 6-dichlorobenzimidazole derivatives as potent androgen receptor antagonists. Bioorganic & Medicinal Chemistry Letters 17(4):955–958
Kimura A, Lee J-H, Lee I-S, Lee H-S, Park K-H, Chiba S et al (2004) Two potent competitive inhibitors discriminating α-glucosidase family I from family II. Carbohydrate Research 339(6):1035–1040
Zawawi NKNA, Taha M, Ahmat N, Ismail NH, Wadood A, Rahim F (2017) Synthesis, molecular docking studies of hybrid benzimidazole as α-glucosidase inhibitor. Bioorganic Chemistry 70:184–191
Jenuwein T (2006) The epigenetic magic of histone lysine methylation. The FEBS journal 273(14):3121–3135
Albert M, Helin K, editors. Histone methyltransferases in cancer. Seminars in Cell & Developmental Biology; 2010: Elsevier.
Zhang J, Yao D, Jiang Y, Huang J, Yang S, Wang J (2017) Synthesis and biological evaluation of benzimidazole derivatives as the G9a Histone Methyltransferase inhibitors that induce autophagy and apoptosis of breast cancer cells. Bioorganic Chemistry 72:168–181
Christofk HR, Vander Heiden MG, Harris MH, Ramanathan A, Gerszten RE, Wei R et al (2008) The M2 splice isoform of pyruvate kinase is important for cancer metabolism and tumour growth. Nature 452(7184):230
Mazurek S (2011) Pyruvate kinase type M2: a key regulator of the metabolic budget system in tumor cells. The international journal of biochemistry & cell biology 43(7):969–980
Guo C, Linton A, Jalaie M, Kephart S, Ornelas M, Pairish M et al (2013) Discovery of 2-((1H-benzo [d] imidazol-1-yl) methyl)-4H-pyrido [1, 2-a] pyrimidin-4-ones as novel PKM2 activators. Bioorganic & Medicinal Chemistry Letters 23(11):3358–3363
Maji B, Kumar K, Kaulage M, Muniyappa K, Bhattacharya S (2014) Design and Synthesis of new benzimidazole–carbazole conjugates for the stabilization of human telomeric DNA, telomerase inhibition, and their selective action on cancer cells. Journal of Medicinal Chemistry 57(16):6973–6988
Jain AK, Paul A, Maji B, Muniyappa K, Bhattacharya S (2012) Dimeric 1, 3-phenylene-bis (piperazinyl benzimidazole) s: synthesis and structure–activity investigations on their binding with human telomeric G-quadruplex DNA and telomerase inhibition properties. Journal of Medicinal Chemistry 55(7):2981–2993
Chen C-L, Chang D-M, Chen T-C, Lee C-C, Hsieh H-H, Huang F-C et al (2013) Structure-based design, synthesis and evaluation of novel anthra [1, 2-d] imidazole-6, 11-dione derivatives as telomerase inhibitors and potential for cancer polypharmacology. European Journal of Medicinal Chemistry 60:29–41
Shi L, Wu T-T, Wang Z, Xue J-Y, Xu Y-G (2014) Discovery of N-(2-phenyl-1H-benzo [d] imidazol-5-yl) quinolin-4-amine derivatives as novel VEGFR-2 kinase inhibitors. European Journal of Medicinal Chemistry 84:698–707
Carmeliet P (2003) Angiogenesis in health and disease. Nature Medicine 9(6):653
Arcari JT, Beebe JS, Berliner MA, Bernardo V, Boehm M, Borzillo GV et al (2013) Discovery and synthesis of novel 4-aminopyrrolopyrimidine Tie-2 kinase inhibitors for the treatment of solid tumors. Bioorganic & Medicinal Chemistry Letters 23(10):3059–3063
Patrick GL (2013) An introduction to medicinal chemistry: Oxford university press
Rowinsky EK (2003) Signal events: cell signal transduction and its inhibition in cancer. The Oncologist 8(Supplement 3):5-17.
Akhtar W, Khan MF, Verma G, Shaquiquzzaman M, Rizvi MA, Mehdi SH et al (2017) Therapeutic evolution of benzimidazole derivatives in the last quinquennial period. European Journal of Medicinal Chemistry 126:705–753
Liu W-Q, Megale V, Borriello L, Leforban B, Montes M, Goldwaser E et al (2014) Synthesis and structure–activity relationship of non-peptidic antagonists of neuropilin-1 receptor. Bioorganic & Medicinal Chemistry Letters 24(17):4254–4259
Temirak A, Shaker YM, Ragab FA, Ali MM, Ali HI, El Diwani HI (2014) Part I. Synthesis, biological evaluation and docking studies of new 2-furylbenzimidazoles as antiangiogenic agents. European Journal of Medicinal Chemistry 87:868–880
Abdullaziz MA, Abdel-Mohsen HT, El Kerdawy AM, Ragab FA, Ali MM, Abu-bakr SM et al (2017) Design, synthesis, molecular docking and cytotoxic evaluation of novel 2-furybenzimidazoles as VEGFR-2 inhibitors. European Journal of Medicinal Chemistry 136:315–329
Ashok A, Thanukrishnan K, Bhojya Naik HS, Maridu R (2017) Novel aryl-modified benzoylamino-N-(5, 6-dimethoxy-1H-benzoimidazol-2-yl)-heteroamides as potent inhibitors of vascular endothelial growth factor receptors 1 and 2. Journal of Heterocyclic Chemistry 54(3):1949–1956
Liao JJ-L (2007) Molecular recognition of protein kinase binding pockets for design of potent and selective kinase inhibitors. Journal of Medicinal Chemistry 50(3):409–424
Zhang J, Yang PL, Gray NS (2009) Targeting cancer with small molecule kinase inhibitors. Nature reviews Cancer 9(1):28
Zuccotto F, Ardini E, Casale E, Angiolini M (2009) Through the “gatekeeper door”: exploiting the active kinase conformation. Journal of Medicinal Chemistry 53(7):2681–2694
Determann R, Dreher J, Baumann K, Preu L, Jones PG, Totzke F et al (2012) 2-Anilino-4-(benzimidazol-2-yl) pyrimidines–A multikinase inhibitor scaffold with antiproliferative activity toward cancer cell lines. European Journal of Medicinal Chemistry 53:254–263
Akhtar MJ, Siddiqui AA, Khan AA, Ali Z, Dewangan RP, Pasha S et al (2017) Design, synthesis, docking and QSAR study of substituted benzimidazole linked oxadiazole as cytotoxic agents, EGFR and erbB2 receptor inhibitors. European Journal of Medicinal Chemistry 126:853–869
Lelais G, Epple R, Marsilje TH, Long YO, McNeill M, Chen B et al (2016) Discovery of (R, E)-N-(7-Chloro-1-(1-[4-(dimethylamino) but-2-enoyl] azepan-3-yl)-1 H-benzo [d] imidazol-2-yl)-2-methylisonicotinamide (EGF816), a novel, potent, and WT sparing covalent inhibitor of oncogenic (L858R, ex19del) and resistant (T790M) EGFR mutants for the treatment of EGFR mutant non-small-cell lung cancers. Journal of Medicinal Chemistry 59(14):6671–6689
Touat M, Ileana E, Postel-Vinay S, André F, Soria J-C (2015) Targeting FGFR signaling in cancer. Clinical Cancer Research 21(12):2684–2694
Yan W, Wang X, Dai Y, Zhao B, Yang X, Fan J et al (2016) Discovery of 3-(5′-Substituted)-benzimidazole-5-(1-(3, 5-dichloropyridin-4-yl) ethoxy)-1H-indazoles as potent fibroblast growth factor receptor inhibitors: design, synthesis, and biological evaluation. J Med Chem 59(14):6690–6708
Gryshchenko A, Tarnavskiy S, Levchenko K, Bdzhola V, Volynets G, Golub A et al (2016) Design, synthesis and biological evaluation of 5-amino-4-(1H-benzoimidazol-2-yl)-phenyl-1, 2-dihydro-pyrrol-3-ones as inhibitors of protein kinase FGFR1. Bioorganic & Medicinal Chemistry 24(9):2053–2059
Huang H, Bhat A, Woodnutt G, Lappe R (2010) Targeting the ANGPT–TIE2 pathway in malignancy. Nature Reviews Cancer 10(8):575–585
Hammond E, Handley P, Dredge K, Bytheway I (2013) Mechanisms of heparanase inhibition by the heparan sulfate mimetic PG545 and three structural analogues. FEBS open bio 3(1):346–351
Pan W, Miao H-Q, Xu Y-J, Navarro EC, Tonra JR, Corcoran E et al (2006) 1-[4-(1H-Benzoimidazol-2-yl)-phenyl]-3-[4-(1H-benzoimidazol-2-yl)-phenyl]-urea derivatives as small molecule heparanase inhibitors. Bioorganic & Medicinal Chemistry Letters 16(2):409–412
Dempke W, Rie C, Grothey A, Schmoll H-J (2001) Cyclooxygenase-2: a novel target for cancer chemotherapy? Journal of Cancer Research and Clinical Oncology 127(7):411–417
Gupta RA, DuBois RN (2001) Colorectal cancer prevention and treatment by inhibition of cyclooxygenase-2. Nature reviews Cancer 1(1):11
Abdelgawad MA, Bakr RB, Omar HA (2017) Design, synthesis and biological evaluation of some novel benzothiazole/benzoxazole and/or benzimidazole derivatives incorporating a pyrazole scaffold as antiproliferative agents. Bioorganic Chemistry.
Marugán C, Torres R, Lallena MJ (2016) Phenotypic screening approaches to develop aurora kinase inhibitors: drug discovery perspectives. Frontiers in Oncology 5:299
Pollard JR, Mortimore M (2009) Discovery and development of aurora kinase inhibitors as anticancer agents. Journal of Medicinal Chemistry 52(9):2629–2651
Zheng Y, Zheng M, Ling X, Liu Y, Xue Y, An L et al (2013) Design, synthesis, quantum chemical studies and biological activity evaluation of pyrazole–benzimidazole derivatives as potent Aurora A/B kinase inhibitors. Bioorganic & Medicinal Chemistry Letters 23(12):3523–3530
Zhong M, Bui M, Shen W, Baskaran S, Allen DA, Elling RA et al (2009) 2-Aminobenzimidazoles as potent Aurora kinase inhibitors. Bioorganic & Medicinal Chemistry Letters 19(17):5158–5161
Howard S, Berdini V, Boulstridge JA, Carr MG, Cross DM, Curry J et al (2008) Fragment-based discovery of the pyrazol-4-yl urea (AT9283), a multitargeted kinase inhibitor with potent aurora kinase activity. Journal of Medicinal Chemistry 52(2):379–388
Sharma A, Luxami V, Paul K (2015) Purine-benzimidazole hybrids: synthesis, single crystal determination and in vitro evaluation of antitumor activities. European Journal of Medicinal Chemistry 93:414–422
Sherr CJ, Bartek J (2016) Cell cycle–targeted cancer therapies. Annual Review of Cancer Biology (0).
Bartek J, Falck J, Lukas J (2001) CHK2 kinase--a busy messenger. Nature reviews Molecular cell biology 2(12):877
Garcia-Limones C, Lara-Chica M, Jimenez-Jimenez C, Pérez M, Moreno P, Muñoz E et al (2016) CHK2 stability is regulated by the E3 ubiquitin ligase SIAH2. Oncogene 35(33):4289–4301
Song P, Peng P, Han M, Cao X, Ma X, Liu T et al (2014) Design, synthesis and biological evaluation of thienopyridinones as Chk1 inhibitors. Bioorganic & Medicinal Chemistry 22(17):4882–4892
Ni Z-J, Barsanti P, Brammeier N, Diebes A, Poon DJ, Ng S et al (2006) 4-(Aminoalkylamino)-3-benzimidazole-quinolinones as potent CHK-1 inhibitors. Bioorganic & Medicinal Chemistry Letters 16(12):3121–3124
Neff DK, Lee-Dutra A, Blevitt JM, Axe FU, Hack MD, Buma JC et al (2007) 2-Aryl benzimidazoles featuring alkyl-linked pendant alcohols and amines as inhibitors of checkpoint kinase Chk2. Bioorganic & Medicinal Chemistry Letters 17(23):6467–6471
Galal SA, Abdelsamie AS, Shouman SA, Attia YM, Ali HI, Tabll A et al (2017) Part I: Design, synthesis and biological evaluation of novel pyrazole-benzimidazole conjugates as checkpoint kinase 2 (Chk2) inhibitors with studying their activities alone and in combination with genotoxic drugs. European Journal of Medicinal Chemistry 134:392–405
El-Naem SI, El-Nzhawy A, El-Diwani H, Abdel Hamid A (2003) Synthesis of 5-substituted 2-methylbenzimidazoles with anticancer activity. Archiv der Pharmazie 336(1):7–17
Trembley J, Wang G, Unger G, Slaton J, Ahmed K (2009) Protein kinase CK2 in health and disease. Cellular and Molecular Life Sciences 66(11-12):1858–1867
Bian Y, Ye M, Wang C, Cheng K, Song C, Dong M, et al. (2013) Global screening of CK2 kinase substrates by an integrated phosphoproteomics workflow. Scientific Reports 3.
Ruzzene M, Pinna LA (2010) Addiction to protein kinase CK2: a common denominator of diverse cancer cells? Biochimica et Biophysica Acta (BBA)-Proteins and Proteomics 1804(3):499-504.
Schneider CC, Kartarius S, Montenarh M, Orzeszko A, Kazimierczuk Z (2012) Modified tetrahalogenated benzimidazoles with CK2 inhibitory activity are active against human prostate cancer cells LNCaP in vitro. Bioorganic & Medicinal Chemistry 20(14):4390–4396
Pagano MA, Meggio F, Ruzzene M, Andrzejewska M, Kazimierczuk Z, Pinna LA (2004) 2-Dimethylamino-4, 5, 6, 7-tetrabromo-1H-benzimidazole: a novel powerful and selective inhibitor of protein kinase CK2. Biochemical and Biophysical Research Communications 321(4):1040–1044
Andrzejewska M, Pagano MA, Meggio F, Brunati AM, Kazimierczuk Z (2003) Polyhalogenobenzimidazoles: synthesis and their inhibitory activity against casein kinases. Bioorganic & Medicinal Chemistry 11(18):3997–4002
Łukowska-Chojnacka E, Wińska P, Wielechowska M, Poprzeczko M, Bretner M (2016) Synthesis of novel polybrominated benzimidazole derivatives—potential CK2 inhibitors with anticancer and proapoptotic activity. Bioorganic & Medicinal Chemistry 24(4):735–741
Malumbres M (2014) Cyclin-dependent kinases. Genome Biology 15(6):122
Lin R, Connolly PJ, Lu Y, Chiu G, Li S, Yu Y et al (2007) Synthesis and evaluation of pyrazolo [3, 4-b] pyridine CDK1 inhibitors as anti-tumor agents. Bioorganic & Medicinal Chemistry Letters 17(15):4297–4302
Gorgisen G, Gulacar I, Ozes O (2017) The role of insulin receptor substrate (IRS) proteins in oncogenic transformation. Cellular and Molecular Biology 63(1):1–5
Yu H, Rohan T (2000) Role of the insulin-like growth factor family in cancer development and progression. Journal of the National Cancer Institute 92(18):1472–1489
Baserga R (1995) The insulin-like growth factor I receptor: a key to tumor growth? Cancer Research 55(2):249–252
Wittman M, Carboni J, Attar R, Balasubramanian B, Balimane P, Brassil P et al (2005) Discovery of a (1 H-Benzoimidazol-2-yl)-1 H-pyridin-2-one (BMS-536924) inhibitor of insulin-like growth factor I receptor kinase with in vivo antitumor activity. Journal of Medicinal Chemistry 48(18):5639–5643
Liu P, Cheng H, Roberts TM, Zhao JJ (2009) Targeting the phosphoinositide 3-kinase (PI3K) pathway in cancer. Nature reviews Drug discovery 8(8):627
Vanhaesebroeck B, Guillermet-Guibert J, Graupera M, Bilanges B (2010) The emerging mechanisms of isoform-specific PI3K signalling. Nature reviews Molecular cell biology 11(5):329
Van Dort ME, Hong H, Wang H, Nino CA, Lombardi RL, Blanks AE et al (2016) Discovery of bifunctional oncogenic target inhibitors against allosteric mitogen-activated protein kinase (MEK1) and phosphatidylinositol 3-kinase (PI3K). Journal of Medicinal Chemistry 59(6):2512–2522
Certal V, Halley F, Virone-Oddos A, Delorme C, Karlsson A, Rak A et al (2012) Discovery and optimization of new benzimidazole-and benzoxazole-pyrimidone selective PI3Kβ inhibitors for the treatment of phosphatase and TENsin homologue (PTEN)-deficient cancers. Journal of Medicinal Chemistry 55(10):4788–4805
Murarka S, Martín-Gago P, Schultz-Fademrecht C, Al Saabi A, Baumann M, Fansa EK et al (2017) Development of pyridazinone chemotypes targeting the PDEδ prenyl binding site. Chemistry-A European Journal 23(25):6083–6093
Zimmermann G, Schultz-Fademrecht C, Küchler P, Murarka S, Ismail S, Triola G et al (2014) Structure guided design and kinetic analysis of highly potent benzimidazole inhibitors targeting the PDEδ prenyl binding site. Journal of Medicinal Chemistry 57(12):5435–5448
Rewcastle GW, Gamage SA, Flanagan JU, Kendall JD, Denny WA, Baguley BC et al (2013) Synthesis and biological evaluation of novel phosphatidylinositol 3-kinase inhibitors: Solubilized 4-substituted benzimidazole analogs of 2-(difluoromethyl)-1-[4, 6-di (4-morpholinyl)-1, 3, 5-triazin-2-yl]-1H-benzimidazole (ZSTK474). European Journal of Medicinal Chemistry 64:137–147
Mortlock AA, Wilson DM, Kettle JG, Goldberg FW, Foote KM. Selective kinase inhibitors in cancer. Reference Module in Chemistry, Molecular Sciences and Chemical Engineering: Elsevier; 2017.
Marco-Contelles J, Soriano E (2011) Editorial [Hot topic: the medicinal chemistry of hybrid-based drugs targeting multiple sites of action (Guest Editors: Jose Marco-Contelles, & Elena Soriano)]. Current Topics in Medicinal Chemistry 11(22):2714–2715
Shi L, Wu T-T, Wang Z, Xue J-Y, Xu Y-G (2014) Discovery of quinazolin-4-amines bearing benzimidazole fragments as dual inhibitors of c-Met and VEGFR-2. Bioorganic & Medicinal Chemistry 22(17):4735–4744
Renhowe PA, Pecchi S, Shafer CM, Machajewski TD, Jazan EM, Taylor C et al (2008) Design, structure−activity relationships and in vivo characterization of 4-amino-3-benzimidazol-2-ylhydroquinolin-2-ones: a novel class of receptor tyrosine kinase inhibitors. Journal of Medicinal Chemistry 52(2):278–292
Li Y, Tan C, Gao C, Zhang C, Luan X, Chen X et al (2011) Discovery of benzimidazole derivatives as novel multi-target EGFR, VEGFR-2 and PDGFR kinase inhibitors. Bioorganic & Medicinal Chemistry 19(15):4529–4535
McBride CM, Renhowe PA, Heise C, Jansen JM, Lapointe G, Ma S et al (2006) Design and structure–activity relationship of 3-benzimidazol-2-yl-1H-indazoles as inhibitors of receptor tyrosine kinases. Bioorganic & Medicinal Chemistry Letters 16(13):3595–3599
Plagiarism declaration
I hereby declare that this submission is my own work and to best of my knowledge, it contains no material previously published or written by another person, except where due acknowledgement is made. Furthermore, I believe that no contents of this material have been accepted for the award of any other degree or diploma in any other university or tertiary institution.
Funding
Not applicable.
Author information
Authors and Affiliations
Contributions
HA: data collection and integration, prepared the first draft of the manuscript. HM: supervision and manuscript preparation. All authors have read and approved the manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ibrahim, H.A., Refaat, H.M. Versatile mechanisms of 2-substituted benzimidazoles in targeted cancer therapy. Futur J Pharm Sci 6, 41 (2020). https://doi.org/10.1186/s43094-020-00048-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s43094-020-00048-8